XXXXXXXXX NAŘÍZENÍ XXXXXX (XX) 2021/608
xx xxx 14.&xxxx;xxxxx 2021,
xxxxxx se xxxx xxxxxxxxx nařízení (XX) 2019/1793 x&xxxx;xxxxxxxx xxxxxxxxxxxxx xxxxxxxx xxxxxxx x&xxxx;xxxxxxxxxxx opatření xxxxxxxxxxxx xxxxx xxxxxxxx zboží x&xxxx;xxxxxxxx xxxxxxx xxxx xx Xxxx, xxxxxx xx xxxxxxxxx xxxxxxxx Xxxxxxxxxx xxxxxxxxxx x&xxxx;Xxxx (XX) 2017/625 a (ES) x.&xxxx;178/2002
(Xxxx x&xxxx;xxxxxxxx xxx XXX)
XXXXXXXX KOMISE,
s ohledem xx Xxxxxxx o fungování Evropské xxxx,
x&xxxx;xxxxxxx na xxxxxxxx Xxxxxxxxxx xxxxxxxxxx x&xxxx;Xxxx (XX) x.&xxxx;178/2002 xx xxx 28. ledna 2002, xxxxxx xx stanoví xxxxxx xxxxxx x&xxxx;xxxxxxxxx potravinového xxxxx, xxxxxxx xx Xxxxxxxx úřad xxx xxxxxxxxxx potravin x&xxxx;xxxxxxx xxxxxxx týkající xx xxxxxxxxxxx potravin (1), a zejména xx čl. 53 xxxx.&xxxx;1 xxxx. x) uvedeného xxxxxxxx,
x&xxxx;xxxxxxx xx nařízení Xxxxxxxxxx xxxxxxxxxx a Rady (XX) 2017/625 xx xxx 15.&xxxx;xxxxxx&xxxx;2017 x&xxxx;xxxxxxxx xxxxxxxxxx x&xxxx;xxxxxx xxxxxxxx xxxxxxxxxx xxxxxxxxxxx x&xxxx;xxxxx xxxxxxxx xxxxxxxxxxx potravinového x&xxxx;xxxxxxxxxx xxxxx a pravidel xxxxxxxxxx se xxxxxx xxxxxx x&xxxx;xxxxxxx životních xxxxxxxx xxxxxx, xxxxxx xxxxxxx a přípravků xx xxxxxxx rostlin, o změně xxxxxxxx Xxxxxxxxxx xxxxxxxxxx x&xxxx;Xxxx (ES) x.&xxxx;999/2001, (XX) č. 396/2005, (ES) x.&xxxx;1069/2009, (ES) x.&xxxx;1107/2009, (XX) č. 1151/2012, (XX) x.&xxxx;652/2014, (EU) 2016/429 x&xxxx;(XX) 2016/2031, xxxxxxxx Xxxx (ES) x.&xxxx;1/2005 x&xxxx;(XX) x.&xxxx;1099/2009 a směrnic Xxxx 98/58/XX, 1999/74/XX, 2007/43/XX, 2008/119/XX a 2008/120/ES x&xxxx;x&xxxx;xxxxxxx xxxxxxxx Evropského xxxxxxxxxx x&xxxx;Xxxx (ES) x.&xxxx;854/2004 a (ES) x.&xxxx;882/2004, xxxxxxx Rady 89/608/XXX, 89/662/XXX, 90/425/EHS, 91/496/XXX, 96/23/XX, 96/93/XX x&xxxx;97/78/XX x&xxxx;xxxxxxxxxx Xxxx 92/438/XXX (xxxxxxxx x&xxxx;xxxxxxxx xxxxxxxxxx)&xxxx;(2), x&xxxx;xxxxxxx na xx.&xxxx;47 xxxx.&xxxx;2 xxxxx xxxxxxxxxxx xxxx. x), xx.&xxxx;54 xxxx.&xxxx;4 xxxxx xxxxxxxxxxx xxxx. a) a b) x&xxxx;xx.&xxxx;90 první pododstavec xxxx. x), b) x&xxxx;x).
xxxxxxxx k těmto důvodům:
|
(1) |
Prováděcí xxxxxxxx Xxxxxx (XX) 2019/1793&xxxx;(3) xxxxxxx pravidla xxxxxxxx xx xxxxxxxxx xxxxxxxxxxxxx xxxxxxxx kontrol xxx xxxxxx xxxxxxxx xxxxxxxx a krmiv xxxxxx xxx xxxxxxxxxxx xxxxxx x&xxxx;xxxxxxxx třetích xxxx xxxxxxxxx xx xxxxxxx x&xxxx;xxxxxxx X&xxxx;xxxxxxxxx prováděcího xxxxxxxx do Xxxx x&xxxx;xxxxxxxx xxxxxxxx xxx xxxxx určitých xxxxxxxx x&xxxx;xxxxx x&xxxx;xxxxxxxx třetích xxxx xxxxxxxxx xx xxxxxxx x&xxxx;xxxxxxx XX xxxxxxxxx prováděcího xxxxxxxx xx Xxxx x&xxxx;xxxxxx xxxxxx xxxxxxxxxxx xxxxxxxxxx, xxxxxx xxxxxxxxxx, xxxxxxx xxxxxxxxx, pentachlorfenolem a dioxiny x&xxxx;xxxxxxxxxxxxxxx kontaminace. |
|
(2) |
Prováděcí nařízení (XX) 2019/1793 stanoví xxxxxxxxx, xxxxx jde x&xxxx;xxxxxxx xxxxxx xxxxxxxxx xxx vstup zásilek xxxxxxxx x&xxxx;xxxxx uvedených x&xxxx;xxxxxxx II uvedeného xxxxxxxxxxx xxxxxxxx xx Xxxx, x&xxxx;xxxxxxxx xxx xxxxxxxx takového osvědčení x&xxxx;xxxxxxx x&xxxx;xxxxxxxxxxxx podobě. X&xxxx;xxxxxxx x&xxxx;xxxxxxxxxx xxxxxxxxx Xxxxxx (XX) 2019/1715&xxxx;(4) xx systém TRACES xxxxxxx xxxxxxx pro xxxxxx informací o úředních xxxxxxxxxx (IMSOC), xxxxx xxxxxxxx, aby celý xxxxxx xxxxxx xxxxxxxxx xxxxxxxx elektronicky, x&xxxx;xxx xx zabránilo xxxxxx xxxxxxxxx xxxx klamavým xxxxxxxxx v souvislosti x&xxxx;xxxxxxxx xxxxxxxxxxx. Xxxxxxxxx xxxxxxxx (XX) 2019/1793 xxxxx xxxxxxx xxxxxxx xxxxxx xxxxxxxxx, jež xx x&xxxx;xxxxxxx x&xxxx;XXXXXX. |
|
(3) |
Xxxxxxxxx na xxxxxxxxxxx stanovené x&xxxx;xxxxxxxxxx xxxxxxxx (XX) 2019/1793 xxxx x&xxxx;xxxxxxx s požadavky xxxxxxxxxxx x&xxxx;xxxxxxxxxx nařízení Xxxxxx (XX) 2019/628 (5) xxx xxxxxx xxxxxxxxx xxx xxxxx xx Xxxx. Xxxxxxxxx nařízení Xxxxxx (XX) 2020/2235 (6) xxxxxxx x&xxxx;xxxxxxxxx xxxxxxxxx xxxxxxxx (XX) 2019/628 xxx xxx 21.&xxxx;xxxxx&xxxx;2021 x&xxxx;xxxxxxxxx x&xxxx;xxxxxxxxx požadavky xx vzorová xxxxxx xxxxxxxxx xxxxxxxxx x&xxxx;xxxxxxxx xxxxxxxxxx nařízení. |
|
(4) |
Prováděcí xxxxxxxx (XX) 2020/2235 zejména xxxxxxxxx xxxx úředními xxxxxxxxxxx xxxxxxxx x&xxxx;xxxxxxx xxxxxx, elektronickými xxxxxxxx xxxxxxxxxxx vydanými x&xxxx;xxxxxxx x&xxxx;xxxxxxxxx čl. 39 xxxx.&xxxx;1 xxxxxxxxxxx xxxxxxxx (XX) 2019/1715 x&xxxx;xxxxxxxx xxxxxxxxxxx xxxxxxxx x&xxxx;xxxxxxx xxxxxx x&xxxx;xxxxxxxxxx x&xxxx;xxxxxxx XXXXXX x&xxxx;xxxxxxxxxxx x&xxxx;xxxxxx xxxxxxx. Xxxxx xxxx xxxxxxx xxxxxxxxx nařízení xxxxxxx xxxxxxxx xxxxxxxxx na xxxxxx osvědčení xxx xxxxx do Xxxx, xxx xx xxxxxxxxx xxxxxx xxxxxxxx xx xxxxxxxxxxxx xxxxxxxx xxxxxxxx x&xxxx;xxxxx vstupu xx Xxxx. Aby xx xxxxxxx úřední osvědčení xxx xxxxx kategorie xxxxx a zajistil xx xxxxxx x&xxxx;xxxxxx xxxxxxxxx xx xxxxxxxxxxx v úředních xxxxxxxxxxx xxx vstup xx Xxxx xxxxxxxxxxx x&xxxx;xxxxxxxxxx xxxxxxxx (EU) 2020/2235, je vhodné xxxxxx xxxxxx&xxxx;11 xxxxxxxxxxx xxxxxxxx (XX) 2019/1793. |
|
(5) |
Xxxxxx&xxxx;12 xxxxxxxxxxx xxxxxxxx (XX) 2019/1793 stanoví, xx xxxxxxx xxxxxxx x&xxxx;xxxxxxxxx X&xxxx;x&xxxx;XX xxxxx x&xxxx;xxxxxx xxxxxxxxxxxxxx xxxx měsíců xxxxxxxxxx přezkoumávány x&xxxx;xxxxx xxxxxxxxx nové xxxxxxxxx xxxxxxxx xx xxxxx x&xxxx;xxxxxxxxx s právem Xxxx. |
|
(6) |
Xxxxxx x&xxxx;xxxxxxxxx nedávných případů x&xxxx;xxxxxxx potravin oznámených xxxxxxxxxxxxxxx xxxxxxx včasné xxxxxx xxxxxxxxx xxx xxxxxxxxx a krmiva (RASFF), xxxxx xxx zřízen xxxxxxxxx (XX) č. 178/2002, x&xxxx;xxxxxxxxx o úředních xxxxxxxxxx xxxxxxxxxxx xxxxxxxxx státy x&xxxx;xxxxxxxx a krmiv xxxxxx xxx živočišného xxxxxx xxxxxxxxx, xx xxxxxxx x&xxxx;xxxxxxxxx X&xxxx;x&xxxx;XX xxxxxxxxxxx xxxxxxxx (XX) 2019/1793 xx xxxx xxx xxxxxxx. |
|
(7) |
Xxxxx xxxx, vzhledem x&xxxx;xxxxxxx nesouladu s příslušnými xxxxxxxxx xxxxxxxxxxx v právních xxxxxxxxxx Xxxx, xxxxx xxx x&xxxx;xxxxxxxxxxx salmonelami xxxxxxxxx xxx xxxxxxxx xxxxxxxxxx xxxxxxxxxxx xxxxxxxxx xxxxx xxxxx xxxxxxxxxxx xxxxxxxx (XX) 2019/1793 x&xxxx;xxxx 2019 x&xxxx;x&xxxx;xxxxxx xxxxxxxx xxxx 2020, x&xxxx;x&xxxx;xxxxxxxx xxxxx xxxxxxxx x&xxxx;xxxxxxx XXXXX xxxxx xxxxxx xxxxxx je xxxxxx xxxxxx xxxxxxx xxxxxxx xxxxxxxxxx x&xxxx;xxxxxxxxx xxxxxxx x&xxxx;xxxxxxx pepře (Xxxxx xxxxxx) x&xxxx;Xxxxxxxx x&xxxx;20 xx 50&xxxx;%. |
|
(8) |
Xxxxxxxx x&xxxx;xxxxxxx xxxxxxxxx s příslušnými xxxxxxxxx xxxxxxxxxxx x&xxxx;xxxxxxxx xxxxxxxxxx Unie, pokud xxx x&xxxx;xxxxxxxxxxx xxxxxxx xxxxxxxxx xxxxxxxxx při xxxxxxxx xxxxxxxxxx xxxxxxxxxxx xxxxxxxxx státy xxxxx xxxxxxxxxxx xxxxxxxx (EU) 2019/1793 ve xxxxxx xxxxxxxx xxxx 2019 x&xxxx;x&xxxx;xxxxxx xxxxxxxx xxxx 2020, je xxxxxx xxxxxx četnost kontrol xxxxxxxxxx a fyzických xxxxxxx x&xxxx;xxxxxxx xxxx Xxxxxxxx (xxxx než xxxxxx) x&xxxx;Xxxxxxx x&xxxx;10 na 20&xxxx;%. |
|
(9) |
Xxxxxxxx x&xxxx;xxxxxxx xxxxxxxxx x&xxxx;xxxxxxxxxxx xxxxxxxxx stanovenými x&xxxx;xxxxxxxx xxxxxxxxxx Xxxx, xxxxx xxx x&xxxx;xxxxxxxxxxx xxxxxxxxxx zjištěnou xxx xxxxxxxx kontrolách xxxxxxxxxxx xxxxxxxxx státy podle xxxxxxxxxxx nařízení (XX) 2019/1793 ve xxxxxx xxxxxxxx xxxx 2019 x&xxxx;x&xxxx;xxxxxx pololetí roku 2020, x&xxxx;x&xxxx;xxxxxxxx xxxxx xxxxxxxx v systému XXXXX xxxxx xxxxxxx xxxxxxxx xxxx 2020 xx xxxxxx xxxxxx xxxxxxx xxxxxxx xxxxxxxxxx a fyzických xxxxxxx x&xxxx;xxxxxxxxxx olejné x&xxxx;Xxxxx x&xxxx;10 na 50&xxxx;%. |
|
(10) |
Xxxxxx xxxxxxx (Capsicum xxxxxx) x&xxxx;Xxxxxxx xx xxx xxxxxxx v příloze X&xxxx;xxxxxxxxxxx nařízení (XX) 2019/1793 z důvodu xxxxxx xxxxxxxxxxx xxxxxxx xxxxxxxxx. X&xxxx;xxxxxxx xxxxxxx xxxxxx xxxx Xxxxxxxx (xxxxxx xxx sladkých) x&xxxx;Xxxxxxx xxxxxxxxx xxxxx xxxxxxx xx xxxxxxx xxxxxxxx xxxxxxxxxx prostřednictvím xxxxxxx XXXXX v prvním xxxxxxxx xxxx 2020 xx xxxxx xxxxxx xxxxx xxx xxxxxx zdraví x&xxxx;xxxxxxxx xxxxx xxxxxxxxxxx xxxxxxx xxxxxxxxx, což xxxxxxxx xxxxxxxx xxxxxxxxxx xxxxxxxx xxxxxxx. Xxxxxxxxx xxxxxxx týkající xx xxxxxx xxxxxxx (Capsicum xxxxxx) z Turecka xx xxxxx xxxx xxx xxxxxxx xxx, aby xx xxxxxxxxxx xx xxxxxxx papriky xxxx Xxxxxxxx. |
|
(11) |
Xxxxx xxx x&xxxx;xxxxxxxxxx xxxxxxx (xxxxxxxxx) x&xxxx;Xxxx xxxxxxxx x&xxxx;xxxxxxx I prováděcího xxxxxxxx (XX) 2019/1793 x&xxxx;xxxxxx xxxxxx kontaminace xxxxxxx xxxxxxxxx a pokud xxx o sušené xxxxxx x&xxxx;Xxxxxxx xxxxxxx xx xxxxxxx xxxxxxx z důvodu xxxxxx xxxxxxxxxxx ochratoxinem X, z dostupných xxxxxxxxx xx xxxxx xxxxxxxx xxxx 2019 x&xxxx;xxxxx xxxxxxxx roku 2020 xxxxxxx xxxxxxx uspokojivý xxxxxx souladu x&xxxx;xxxxxxxxxxx xxxxxxxxx stanovenými v právních xxxxxxxxxx Xxxx. Xxxxxxxx x&xxxx;xxxx, že zesílené xxxxxx xxxxxxxx již xxxxx xxxxxx u uvedených xxxxxxx xxxxxxxxxx, xxxx xx být xxxxxxx x&xxxx;xxxxxxx I prováděcího xxxxxxxx (XX) 2019/1793 xxxxxxxx xx xxxxxxxxx xxxxxxx xxxxxxx. |
|
(12) |
Xxxxx xxx x&xxxx;xxxxxxxxxx xxxxxxx z Brazílie xxxxxxxx x&xxxx;xxxxxxx XX xxxxxxxxxxx xxxxxxxx (XX) 2019/1793 x&xxxx;xxxxxx xxxxxx xxxxxxxxxxx xxxxxxxxxx, xxxxx ke xxxxxxx četnosti případů xxxxxxxxx x&xxxx;xxxxxxxxxxx xxxxxxxxx xxxxxxxx xxxxxxxx Xxxx, xxx xxxx zjištěny xxx úředních xxxxxxxxxx xxxxxxxxxxx xxxxxxxxx xxxxx xx druhém pololetí xxxx 2019, a v prvním xxxxxxxx xxxx 2020 xxxxxxx xx xxxxx xxxxxx. Xx xxxxx xxxxxx odstranit xxxxxxx xxxxxxxx xx xxxxxxxxxx xxxxxx z Brazílie x&xxxx;xxxxxxx XX xxxxxxxxxxx xxxxxxxx (XX) 2019/1793 x&xxxx;xxxxxxx xx xx přílohy X&xxxx;xxxxxxxxx prováděcího nařízení x&xxxx;xxxxxxxx xxxxxxx xxxxxxx xxxxxxxxxx a fyzických xxxxxxx xx 10&xxxx;%. |
|
(13) |
Xxxxx jde x&xxxx;xxxxxxxxxx xxxxxxx z Číny xxxxxxxx v příloze XX xxxxxxxxxxx nařízení (XX) 2019/1793 z důvodu xxxxxx xxxxxxxxxxx xxxxxxxxxx, xxxxx xx snížení četnosti xxxxxxx xxxxxxxxx x&xxxx;xxxxxxxxxxx xxxxxxxxx xxxxxxxx předpisů Xxxx, xxx xxxx xxxxxxxx xxx xxxxxxxx xxxxxxxxxx xxxxxxxxxxx členskými xxxxx xx xxxxxx xxxxxxxx xxxx 2019 x&xxxx;x&xxxx;xxxxxx xxxxxxxx xxxx 2020. Je proto xxxxxx xxxxxxxxx xxxxxxx xxxxxxxx se podzemnice xxxxxx x&xxxx;Xxxx z přílohy XX xxxxxxxxxxx xxxxxxxx (XX) 2019/1793 a zařadit xx do přílohy X&xxxx;xxxxxxxxx prováděcího xxxxxxxx x&xxxx;xxxxxxxx xxxxxxx kontrol xxxxxxxxxx a fyzických xxxxxxx xx 10&xxxx;%. S ohledem xx objem xxxxxxx x&xxxx;xxxxx xxxxxxxxx xx xxxx četnost xxxxxxxxxx x&xxxx;xxxxxxxxx odpovídající xxxxxx xxxxxxxxx. |
|
(14) |
Xxxxx xxx x&xxxx;xxxxxxx xxxxxx x&xxxx;Xxxxxxx uvedené x&xxxx;xxxxxxx II xxxxxxxxxxx xxxxxxxx (XX) 2019/1793 x&xxxx;xxxxxx rizika xxxxxxxxxxx xxxxxxxxxx, xxxxx xx xxxxxxx xxxxxxxx xxxxxxx xxxxxxxxx x&xxxx;xxxxxxxxxxx xxxxxxxxx xxxxxxxx předpisů Xxxx, xxx xxxx zjištěny xxx úředních xxxxxxxxxx xxxxxxxxxxx xxxxxxxxx xxxxx xx xxxxxx pololetí xxxx 2019 a v prvním xxxxxxxx xxxx 2020. Xx xxxxx xxxxxx xxxxxxxxx xxxxxxx xxxxxxxx xx xxxxxxxxx ořechů x&xxxx;Xxxx x&xxxx;xxxxxxx II xxxxxxxxxxx nařízení (EU) 2019/1793 a zařadit ji xx xxxxxxx I uvedeného xxxxxxxxxxx xxxxxxxx x&xxxx;xxxxxxxx xxxxxxx xxxxxxx totožnosti x&xxxx;xxxxxxxxx xxxxxxx xx 5&xxxx;%. X&xxxx;xxxxxxx xx xxxxx xxxxxxx x&xxxx;xxxxx xxxxxxxxx xx tato xxxxxxx dostatečná x&xxxx;xxxxxxxxx xxxxxxxxxxxx xxxxxx sledování. |
|
(15) |
Potraviny xxxxxxxxxx listy pepře xxxxxxxxxx (Xxxxx xxxxx) xxxx x&xxxx;xxxx xxxxxxxxxxx xxxxxxxxxxx nebo xxxxxxxx x&xxxx;Xxxxxxxxxx xxxx xxxxxxx x&xxxx;xxxxxxx IIa prováděcího xxxxxxxx (EU) 2019/1793 x&xxxx;xxxxxx xxxxxx xxxxxxxxxxx xxxxxxxxxxx. Xxxxx xxxxxx xxxxxxx do Unie xx proto xx xxxxxx 2014 zakázán. Xxxxxxxxx xxxxxxx xxxxxxx xxxxxx xxx, xx xxx 27.&xxxx;xxxxxxxx&xxxx;2020 xxxxxxxxx xxxx xxxxx xxxx xxxxxxxxxx opatření týkající xx xxxxx xxxx xxxxxxxxx xxxxxxx a Komise xxx xxxxxxxxxxx xxxx xxxxxxxxxx. X&xxxx;xxxxxxxxxx xx xxxx xxxxxxxxx xx xxxxx vhodné odstranit xxxxxxx xxxxxxxx xx xxxxxxxx xxxxxxxxxxxx listy xxxxx xxxxxxxxxx (Xxxxx xxxxx) xxxx x&xxxx;xxxx xxxxxxxxxxxxx xxxxxxxxxxxxx xxxx xxxxxxxxxx z Bangladéše z přílohy XXx xxxxxxxxxxx xxxxxxxx (XX) 2019/1793 x&xxxx;xxxxxxx xx do přílohy XX xxxxxxxxx xxxxxxxxxxx xxxxxxxx a stanovit četnost xxxxxxx xxxxxxxxxx x&xxxx;xxxxxxxxx xxxxxxx na 50&xxxx;%. |
|
(16) |
Xxx xxxx xxxxxxxxx účinná xxxxxxx xxxx xxxxxxx xxxxxxxxxxx xxxxxx xxxxxxxxxxxxx x&xxxx;xxxxxxxxxxxxxxx nebo chemické xxxxxxxxxxx xxxxxxxxxx semen, xx xxxxxxxxx odkazujících xx „xxx XX“ x&xxxx;xxxxxxxxx v přílohách X&xxxx;x&xxxx;XX xxxxxxxxxxx nařízení (XX) 2019/1793 xx xxx xx být do xxxxx s odkazem xx „xxxxxxxx xxxxxx (xxxxxxxxx)“ xxxxxxx xxx XX xxx pražená xxxxxxxx xxxxxx. |
|
(17) |
X&xxxx;xxxxx II xxxxxxxxx xxxxxxxx xxxxxxxxx x&xxxx;xxxxxxx XX prováděcího xxxxxxxx (XX) 2019/1793 se xxxxxxx xxxxxxxxx xxxxxxxxx, xxx xxxxxxxxxxx xxxxxxx xxxxxxxx při vyplňování xxxxxxxxx. X&xxxx;xxxxx xxxxxxxxx xxxxxx jistoty xx xxxx xxx xxxxxxxxx, xx xxxxxxxxx xxxxxxxxx xxxxxxxx se xxxxxxxx xxxx xxxxx xxxxxx xxx xxxxxxxxxxx xxxxxx xxxxx xxxxxxxxx xxxx xxx xxxxx xxxxxxxxx, xxxxx xx xxxxxx xxxxxxxxx povinné xxxxx xx.&xxxx;11 odst. 1 xxxxxxxxxxx xxxxxxxx (XX) 2019/1793 xx xxxxxxx x&xxxx;xxxxxxxx XX uvedeného xxxxxxxxxxx xxxxxxxx. |
|
(18) |
Xxxxxxxxx xxxxxxxx (XX) 2019/1793 by proto xxxx xxx xxxxxxxxxxxxx xxxxxxxx změněno. X&xxxx;xxxxx xxxxxxxxx xxxxxxxxxxxxxxx x&xxxx;xxxxxxxxxxxx xx xxxxxx xxxxxxxx xxxxxxx X, XX, XXx a IV xxxxxxxxx xxxxxxxxxxx nařízení v jejich xxxxxxxx. |
|
(19) |
Xxxxxxxx k tomu, že xx prováděcí nařízení (XX) 2020/2235 použije xxx dne 21. dubna 2021, xxx xx xx xx.&xxxx;1 odst. 1 xxxxxx xxxxxxxx xxxxxx xxxxxx xx xxxxxxxxx data. |
|
(20) |
Opatření xxxxxxxxx tímto nařízením xxxx x&xxxx;xxxxxxx xx xxxxxxxxxxx Xxxxxxx xxxxxx xxx rostliny, xxxxxxx, xxxxxxxxx x&xxxx;xxxxxx, |
XXXXXXX XXXX XXXXXXXX:
Článek 1
Změny prováděcího nařízení (XX) 2019/1793
Xxxxxxxxx xxxxxxxx (XX) 2019/1793 se xxxx xxxxx:
|
1) |
Xxxxxx&xxxx;11 xx xxxxxxxxx xxxxx: „Xxxxxx&xxxx;11 Xxxxxx xxxxxxxxx 1.&xxxx;&xxxx;&xxxx;Xxxxx xxxxxxx xxxxxxxx a krmiv xxxxxxxxx x&xxxx;xxxxxxx XX xxxx xxx xxxxxxxxxxx xxxxxxx xxxxxxxxxx x&xxxx;xxxxxxx xx xxxxxx xxxxxxxx x&xxxx;xxxxxxx XX (xxxx xxx „úřední osvědčení“). 2. Úřední xxxxxxxxx xxxx xxxxxxxx xxxx xxxxxxxxx:
3. Odchylně xx xxxx.&xxxx;2 xxxx. x) xxxx členský xxxx xxxxxxxxx x&xxxx;xxx, xxx byla xxxxxx xxxxxxxxx vyhotovena x&xxxx;xxxxx xxxxxxx xxxxxx Xxxx x, xx-xx xx xxxxxxxx, xxxxxxxx xxxxxxxx xxxxxxxxx. 4.&xxxx;&xxxx;&xxxx;Xxxxxx x&xxxx;xxxxxxx xxxx xxx reliéfní xxxx xxxxxxxxx, na něž xxxxxxxx odst. 2 xxxx. x), jsou v jiné xxxxx xxx barva xxxxx. 5.&xxxx;&xxxx;&xxxx;Xxxxxxxxxx xxxx.&xxxx;2 písm. x) xx x) x&xxxx;xxxxxxxx 4 xx xxxxxxxxx na xxxxxxxxxxxx xxxxxx osvědčení vydaná x&xxxx;xxxxxxx x&xxxx;xxxxxxxxx xx.&xxxx;39 xxxx.&xxxx;1 xxxxxxxxxxx xxxxxxxx Xxxxxx (XX) 2019/1715 (*1). 6.&xxxx;&xxxx;&xxxx;Xxxxxxxxxx xxxx.&xxxx;2 xxxx. x), x) x&xxxx;x) xx xxxxxxxxx xx úřední xxxxxxxxx xxxxxx x&xxxx;xxxxxxx xxxxxx x&xxxx;xxxxxxxx x&xxxx;xxxxxxx XXXXXX x&xxxx;x&xxxx;xxxxxx xxxxxxx xxxxxxxxx. 7.&xxxx;&xxxx;&xxxx;Xxxxxxxxx xxxxxx xxxxx xxxxx náhradní xxxxxx xxxxxxxxx xxxxx v souladu x&xxxx;xxxxxxxx xxxxxxxxxxx x&xxxx;xxxxxx&xxxx;6 xxxxxxxxxxx xxxxxxxx Xxxxxx (XX) 2020/2235 (*2). 8.&xxxx;&xxxx;&xxxx;Xxxxxx osvědčení xx xxxxxx podle xxxxxxxx uvedených x&xxxx;xxxxxxx XX. (*1)&xxxx;&xxxx;Xxxxxxxxx xxxxxxxx Komise (XX) 2019/1715 xx xxx 30.&xxxx;xxxx&xxxx;2019, xxxxxx xx xxxxxxx xxxxxxxx xxx fungování systému xxx správu xxxxxxxxx x&xxxx;xxxxxxxx xxxxxxxxxx x&xxxx;xxxx xxxxxxxxxxx xxxxxx („nařízení x&xxxx;XXXXX“) (Úř. věst. L 261, 14.10.2019, s. 37)." (*2)&xxxx;&xxxx;Xxxxxxxxx xxxxxxxx Xxxxxx (EU) 2020/2235 xx xxx 16. prosince 2020, xxxxxx xx xxxxxxx xxxxxxxxx xxxxxxxx x&xxxx;xxxxxxxxx Xxxxxxxxxx xxxxxxxxxx a Rady (XX) 2016/429 x&xxxx;(XX) 2017/625, xxxxx xxx x&xxxx;xxxxxxx veterinární xxxxxxxxx, xxxxxxx xxxxxx osvědčení x&xxxx;xxxxxxx veterinární/úřední osvědčení xxx vstup xxxxxxx xxxxxxxx xxxxxxxxx xxxxxx x&xxxx;xxxxx xx Xxxx x&xxxx;xxxxxx xxxxxxxxxxxx x&xxxx;xxxxx Xxxx a o úřední xxxxxxxxxxx xxxxxxxx se xxxxxx xxxxxxxxx, x&xxxx;xxxxxx se xxxxxxx xxxxxxxx (XX) x.&xxxx;599/2004, prováděcí nařízení (XX) x.&xxxx;636/2014 a (EU) 2019/628, směrnice 98/68/XX x&xxxx;xxxxxxxxxx 2000/572/ES, 2003/779/ES x&xxxx;2007/240/XX (Úř. věst. L 442, 30.12.2020, s. 1).“." |
|
2) |
Xxxxxxx X, XX, XXx x&xxxx;XX xx nahrazují xxxxxx xxxxxxxx x&xxxx;xxxxxxx xxxxxx xxxxxxxx. |
Článek 2
Vstup v platnost a použitelnost
Toto xxxxxxxx xxxxxxxx x&xxxx;xxxxxxxx xxxxxxxx xxxx xx xxxxxxxxx x&xxxx;Xxxxxxx věstníku Xxxxxxxx xxxx.
Xxxxxxxxxx xx.&xxxx;1 xxxx.&xxxx;1 se xxxxxxx xxx xxx 21.&xxxx;xxxxx&xxxx;2021.
Xxxx xxxxxxxx je xxxxxxx x&xxxx;xxxxx xxxxxxx x&xxxx;xxxxx xxxxxxxxxx xx xxxxx členských xxxxxxx.
X&xxxx;Xxxxxxx xxx 14. dubna 2021.
Xx Xxxxxx
xxxxxxxxxxx
Xxxxxx XXX XXX XXXXX
(1)&xxxx;&xxxx;Xx. xxxx. X&xxxx;31, 1.2.2002, x.&xxxx;1.
(2)&xxxx;&xxxx;Xx. xxxx. X&xxxx;95, 7.4.2017, x.&xxxx;1.
(3)&xxxx;&xxxx;Xxxxxxxxx xxxxxxxx Xxxxxx (XX) 2019/1793 xx dne 22.&xxxx;xxxxx&xxxx;2019 o dočasném zintenzivnění xxxxxxxx kontrol a mimořádných xxxxxxxx xxxxxxxxxxxx vstup xxxxxxxx xxxxx z určitých xxxxxxx xxxx xx Xxxx, xxxxxx se xxxxxxxxx xxxxxxxx Evropského xxxxxxxxxx x&xxxx;Xxxx (XX) 2017/625 a (ES) x.&xxxx;178/2002 x&xxxx;xxxxxx xx xxxxxxx xxxxxxxx Xxxxxx (XX) x.&xxxx;669/2009, (XX) x.&xxxx;884/2014, (XX) 2015/175, (XX) 2017/186 x&xxxx;(XX) 2018/1660 (Xx. věst. X&xxxx;277, 29.10.2019, s. 89).
(4) Prováděcí xxxxxxxx Xxxxxx (XX) 2019/1715 xx dne 30.&xxxx;xxxx&xxxx;2019, xxxxxx se xxxxxxx xxxxxxxx xxx xxxxxxxxx xxxxxxx xxx xxxxxx xxxxxxxxx o úředních kontrolách x&xxxx;xxxx systémových složek („xxxxxxxx x&xxxx;XXXXX“) (Xx. xxxx. L 261, 14.10.2019, x.&xxxx;37).
(5)&xxxx;&xxxx;Xxxxxxxxx xxxxxxxx Komise (XX) 2019/628 ze xxx 8.&xxxx;xxxxx&xxxx;2019 x&xxxx;xxxxxxxxx xxxxxxxx osvědčeních xxx xxxxxxx zvířata x&xxxx;xxxxx x&xxxx;x&xxxx;xxxxx xxxxxxxx (XX) x.&xxxx;2074/2005 x&xxxx;xxxxxxxxxxx xxxxxxxx (XX) 2016/759, xxxxx xxx x&xxxx;xxxx vzorová xxxxxxxxx (Xx. věst. X&xxxx;131, 17.5.2019, x.&xxxx;101).
(6)&xxxx;&xxxx;Xxxxxxxxx xxxxxxxx Xxxxxx (XX) 2020/2235 xx xxx 16.&xxxx;xxxxxxxx&xxxx;2020, xxxxxx xx xxxxxxx xxxxxxxxx xxxxxxxx x&xxxx;xxxxxxxxx Xxxxxxxxxx xxxxxxxxxx x&xxxx;Xxxx (EU) 2016/429 x&xxxx;(XX) 2017/625, xxxxx xxx x&xxxx;xxxxxxx xxxxxxxxxxx xxxxxxxxx, xxxxxxx xxxxxx xxxxxxxxx a vzorová xxxxxxxxxxx/xxxxxx xxxxxxxxx xxx xxxxx xxxxxxx xxxxxxxx xxxxxxxxx xxxxxx a zboží xx Xxxx a jejich xxxxxxxxxxxx x&xxxx;xxxxx Xxxx a o úřední xxxxxxxxxxx xxxxxxxx xx xxxxxx xxxxxxxxx, a kterým xx xxxxxxx xxxxxxxx (XX) x.&xxxx;599/2004, prováděcí xxxxxxxx (XX) č. 636/2014 x&xxxx;(XX) 2019/628, směrnice 98/68/XX x&xxxx;xxxxxxxxxx 2000/572/ES, 2003/779/XX x&xxxx;2007/240/XX (Xx. xxxx. X&xxxx;442, 30.12.2020, x.&xxxx;1).
PŘÍLOHA X
Xxxxxxxxx x&xxxx;xxxxxx xxxxxx xxx živočišného xxxxxx, xxxxx xxxxxxxxx x&xxxx;xxxxxxxx xxxxxxx zemí x&xxxx;xxxxxxxxx dočasnému zintenzivnění xxxxxxxx xxxxxxx xx xxxxxxxxxxxx xxxxxxxx xxxxxxxx x&xxxx;xx xxxxxxxxxxx xxxxxxx
|
Xxxxx |
Xxxxxxxxx x&xxxx;xxxxxx (xxxxxxxxx použití) |
Kód KN &xxxx;(1) |
Xxxxxxx TARIC |
Země xxxxxx |
Xxxxxx |
Xxxxxxx xxxxxxx xxxxxxxxxx a fyzických xxxxxxx (%) |
||||||
|
1 |
|
|
Bolívie (XX) |
Xxxxxxxxxx |
50 |
|||||||
|
|
|||||||||||
|
|
|||||||||||
|
|
|||||||||||
|
|
|||||||||||
(Xxxxxxxxx x&xxxx;xxxxxx) |
|
20 |
||||||||||
|
2 |
(Xxxxxxxxx – xxxxxxxx xxx xxxxxxx) |
xx&xxxx;0904&xxxx;11&xxxx;00 |
10 |
Xxxxxxxx (XX) |
Xxxxxxxxx &xxxx;(2) |
50 |
||||||
|
3 |
|
|
Xxxxxxxx (XX) |
Xxxxxxxxxx |
10 |
|||||||
|
|
|||||||||||
|
|
|||||||||||
|
|
|||||||||||
|
|
|||||||||||
(Xxxxxxxxx x&xxxx;xxxxxx) |
|
20 |
||||||||||
|
4 |
|
|
Xxxx (XX) |
Xxxxxxxxxx |
10 |
|||||||
|
|
|||||||||||
|
|
|||||||||||
|
|
|||||||||||
|
|
|||||||||||
(Xxxxxxxxx x&xxxx;xxxxxx) |
|
20 |
||||||||||
|
5 |
Xxxxxx xxxxxxx (Xxxxxxxx xxxxxx) (Xxxxxxxxx – xxxxxx nebo xxxxx) |
xx&xxxx;0904&xxxx;22&xxxx;00 |
11 |
Xxxx (XX) |
Xxxxxxxxx &xxxx;(6) |
20 |
||||||
|
6 |
Xxx, xxx aromatizovaný (Potraviny) |
0902 |
Xxxx (XX) |
Xxxxxxx xxxxxxxxx &xxxx;(3) &xxxx;(7) |
20 |
|||||||
|
7 |
Xxxxx (Xxxxxxx melongena) (Potraviny – čerstvé nebo xxxxxxxx) |
0709 30 00 |
Dominikánská republika (XX) |
Xxxxxxx xxxxxxxxx &xxxx;(3) |
20 |
|||||||
|
8 |
|
|
Xxxxxxxxxxxx xxxxxxxxx (DO) |
Rezidua xxxxxxxxx (3) &xxxx;(8) |
50 |
|||||||
|
|
20 |
||||||||||
|
ex 0710 80 59 |
20 |
|||||||||||
(Xxxxxxxxx – xxxxxxx, xxxxxxxx xxxx xxxxxxxx) |
|
10 |
||||||||||
|
ex 0710 22 00 |
10 |
|||||||||||
|
9 |
|
|
Xxxxx (XX) |
Xxxxxxx pesticidů &xxxx;(3) &xxxx;(9) |
20 |
|||||||
(Xxxxxxxxx – xxxxxxx, chlazené xxxx xxxxxxxx) |
|
20 |
||||||||||
|
xx&xxxx;0710&xxxx;80&xxxx;59 |
20 |
|||||||||||
|
10 |
Xxxxxxxx xxxxxx (Xxxxxxxxx) |
|
Etiopie (XX) |
Xxxxxxxxx &xxxx;(2) |
50 |
|||||||
|
40 |
|||||||||||
|
40 |
|||||||||||
|
11 |
|
|
Gruzie (GE) |
Aflatoxiny |
50 |
|||||||
|
|
|||||||||||
|
|
40 |
||||||||||
(Potraviny) |
|
30 |
||||||||||
|
ex 2008 19 95 ; |
20 |
|||||||||||
|
xx&xxxx;2008&xxxx;19&xxxx;99 |
30 |
|||||||||||
|
12 |
Xxxxxxx xxxx (Xxxxxxxxx) |
1511 10 90 ; 1511&xxxx;90&xxxx;11 ; |
Xxxxx (GH) |
Barviva Xxxxx &xxxx;(10) |
50 |
|||||||
|
xx&xxxx;1511&xxxx;90&xxxx;19 ; |
90 |
|||||||||||
|
1511&xxxx;90&xxxx;99 |
||||||||||||
|
13 |
Xxxxx curry (Xxxxxxx/Xxxxxxx koenigii) (Potraviny – xxxxxxx, xxxxxxxx, xxxxxxxx xxxx sušené) |
xx&xxxx;1211&xxxx;90&xxxx;86 |
10 |
Xxxxx (XX) |
Xxxxxxx xxxxxxxxx &xxxx;(3) &xxxx;(11) |
50 |
||||||
|
14 |
Xxxx (Xxxxxxxxx – xxxxxxx, xxxxxxxx xxxx zmrazené) |
xx&xxxx;0709&xxxx;99&xxxx;90 ; |
20 |
Xxxxx (IN) |
Rezidua xxxxxxxxx &xxxx;(3) (12) |
10 |
||||||
|
ex 0710 80 95 |
30 |
|||||||||||
|
15 |
Fazole (Xxxxx xxx., Xxxxxxxxx xxx.) (Xxxxxxxxx – čerstvé xxxx xxxxxxxx) |
0708&xxxx;20 |
Xxxx (XX) |
Xxxxxxx xxxxxxxxx&xxxx;(3) |
10 |
|||||||
|
16 |
Xxxxx celer (Xxxxx xxxxxxxxxx) (Xxxxxxxxx – xxxxxxx, čerstvé xxxx xxxxxxxx) |
xx&xxxx;0709&xxxx;40&xxxx;00 |
20 |
Xxxxxxxx (XX) |
Xxxxxxx xxxxxxxxx (3) (13) |
50 |
||||||
|
17 |
Fazole (Vigna xxxxxxxxxxx ssp. sesquipedalis, Xxxxx xxxxxxxxxxx xxx. xxxxxxxxxxx) (Xxxxxxxxx – xxxxxxxx, xxxxxxx, xxxxxxxx xxxx xxxxxxxx) |
ex 0708 20 00 ; |
10 |
Xxxxxxxx (XX) |
Xxxxxxx xxxxxxxxx (3) &xxxx;(14) |
50 |
||||||
|
xx&xxxx;0710&xxxx;22&xxxx;00 |
10 |
|||||||||||
|
18 |
Xxxxxxx (Xxxxxxxx xxxx xxx. rapa) (Potraviny – xxxxxxxxxx nebo konzervované x&xxxx;xxxx nebo xxxxxxxx xxxxxx) |
ex 2001 90 97 |
11; 19 |
Xxxxxxx (XX) |
Xxxxxxxx B |
50 |
||||||
|
19 |
Vodnice (Brassica xxxx xxx. rapa) (Potraviny – xxxxxxxxxx xxxx xxxxxxxxxxxx xx xxxxxx xxxxxx nebo xxxxxxxx xxxxxxxxx, xxxxxxxxxx) |
ex 2005 99 80 |
93 |
Libanon (XX) |
Xxxxxxxx B |
50 |
||||||
|
20 |
Papriky (Xxxxxxxx xxx.) (xxxxxx nebo xxxx xxx xxxxxx) (Xxxxxxxxx – xxxxxx, pražené, xxxxxx xxxx xxxxx) |
0904 21 10 ; |
Xxx Xxxxx (XX) |
Xxxxxxxxxx |
50 |
|||||||
|
xx&xxxx;0904&xxxx;21&xxxx;90 ; |
20 |
|||||||||||
|
xx&xxxx;0904&xxxx;22&xxxx;00 ; |
11; 19 |
|||||||||||
|
ex 2005 99 10 ; |
10; 90 |
|||||||||||
|
xx&xxxx;2005&xxxx;99&xxxx;80 |
94 |
|||||||||||
|
21 |
|
|
Xxxxxxxxxx (XX) |
Xxxxxxxxxx |
50 |
|||||||
|
|
|||||||||||
|
|
|||||||||||
|
|
|||||||||||
|
|
|||||||||||
(Xxxxxxxxx x&xxxx;xxxxxx) |
|
20 |
||||||||||
|
22 |
Xxxxxxxxx (Xxxxxxxxxx xxxxxxxxxxxxx) (Xxxxxxxxx – čerstvé) |
xx&xxxx;0810&xxxx;90&xxxx;20 |
20 |
Xxxxxxxx (XX) |
Xxxxxxx xxxxxxxxx &xxxx;(3) |
20 |
||||||
|
23 |
Xxxxxxxx xxxxxx (Xxxxxxxxx) |
|
Xxxxxxx (XX) |
Xxxxxxxxx &xxxx;(2) |
50 |
|||||||
|
40 |
|||||||||||
|
40 |
|||||||||||
|
24 |
Xxxxx xxxxxx (Xxxxxxxxx) |
0910 91 10 ; 0910&xxxx;91&xxxx;90 |
Xxxxxxxx (PK) |
Aflatoxiny |
50 |
|||||||
|
25 |
Melounová (egusi, Xxxxxxxxx xxx.) xxxxx x&xxxx;xxxxxxxx z nich vyrobené (Potraviny) |
xx&xxxx;1207&xxxx;70&xxxx;00 ; |
10 |
Xxxxxx Leone (XX) |
Xxxxxxxxxx |
50 |
||||||
|
xx&xxxx;1208&xxxx;90&xxxx;00 ; |
10 |
|||||||||||
|
ex 2008 99 99 |
50 |
|||||||||||
|
26 |
|
|
Senegal (SN) |
Aflatoxiny |
50 |
|||||||
|
|
|||||||||||
|
|
|||||||||||
|
|
|||||||||||
|
|
|||||||||||
(Xxxxxxxxx x&xxxx;xxxxxx) |
|
20 |
||||||||||
|
27 |
Xxxxxxx (Brassica rapa xxx. xxxx) (Xxxxxxxxx – xxxxxxxxxx xxxx xxxxxxxxxxxx x&xxxx;xxxx xxxx kyselině xxxxxx) |
xx&xxxx;2001&xxxx;90&xxxx;97 |
11; 19 |
Xxxxx (XX) |
Xxxxxxxx X |
50 |
||||||
|
28 |
Xxxxxxx (Brassica xxxx xxx. xxxx) (Xxxxxxxxx – připravené xxxx xxxxxxxxxxxx ve xxxxxx xxxxxx xxxx xxxxxxxx xxxxxxxxx, xxxxxxxxxx) |
xx&xxxx;2005&xxxx;99&xxxx;80 |
93 |
Xxxxx (XX) |
Xxxxxxxx B |
50 |
||||||
|
29 |
Papriky (xxxx xxx xxxxxx) (Capsicum xxx.) (Xxxxxxxxx – xxxxxxx, xxxxxxxx nebo zmrazené) |
xx&xxxx;0709&xxxx;60&xxxx;99 ; |
20 |
Xxxxxxx (XX) |
Xxxxxxx xxxxxxxxx (3) (15) |
20 |
||||||
|
ex 0710 80 59 |
20 |
|||||||||||
|
30 |
|
|
Turecko (XX) |
Xxxxxxxxxx |
5 |
|||||||
|
|
|||||||||||
|
|
70 |
||||||||||
|
xx&xxxx;0813&xxxx;50&xxxx;91 ; |
70 |
|||||||||||
|
xx&xxxx;0813&xxxx;50&xxxx;99 |
70 |
|||||||||||
|
|
70 |
||||||||||
|
xx&xxxx;2007&xxxx;10&xxxx;99 ; |
40 |
|||||||||||
|
xx&xxxx;2007&xxxx;99&xxxx;39 ; |
05; 06 |
|||||||||||
|
xx&xxxx;2007&xxxx;99&xxxx;50 ; |
33 |
|||||||||||
|
ex 2007 99 97 |
23 |
|||||||||||
|
|
30 |
||||||||||
|
xx&xxxx;2008&xxxx;19&xxxx;19 ; |
30 |
|||||||||||
|
xx&xxxx;2008&xxxx;19&xxxx;92 ; |
30 |
|||||||||||
|
xx&xxxx;2008&xxxx;19&xxxx;95 ; |
20 |
|||||||||||
|
xx&xxxx;2008&xxxx;19&xxxx;99 ; |
30 |
|||||||||||
|
ex 2008 97 12 ; |
15 |
|||||||||||
|
xx&xxxx;2008&xxxx;97&xxxx;14 ; |
15 |
|||||||||||
|
ex 2008 97 16 ; |
15 |
|||||||||||
|
xx&xxxx;2008&xxxx;97&xxxx;18 ; |
15 |
|||||||||||
|
xx&xxxx;2008&xxxx;97&xxxx;32 ; |
15 |
|||||||||||
|
xx&xxxx;2008&xxxx;97&xxxx;34 ; |
15 |
|||||||||||
|
xx&xxxx;2008&xxxx;97&xxxx;36 ; |
15 |
|||||||||||
|
xx&xxxx;2008&xxxx;97&xxxx;38 ; |
15 |
|||||||||||
|
ex 2008 97 51 ; |
15 |
|||||||||||
|
ex 2008 97 59 ; |
15 |
|||||||||||
|
xx&xxxx;2008&xxxx;97&xxxx;72 ; |
15 |
|||||||||||
|
ex 2008 97 74 ; |
15 |
|||||||||||
|
xx&xxxx;2008&xxxx;97&xxxx;76 ; |
15 |
|||||||||||
|
xx&xxxx;2008&xxxx;97&xxxx;78 ; |
15 |
|||||||||||
|
ex 2008 97 92 ; |
15 |
|||||||||||
|
xx&xxxx;2008&xxxx;97&xxxx;93 ; |
15 |
|||||||||||
|
xx&xxxx;2008&xxxx;97&xxxx;94 ; |
15 |
|||||||||||
|
xx&xxxx;2008&xxxx;97&xxxx;96 ; |
15 |
|||||||||||
|
xx&xxxx;2008&xxxx;97&xxxx;97 ; |
15 |
|||||||||||
|
xx&xxxx;2008&xxxx;97&xxxx;98 |
15 |
|||||||||||
|
|
40 |
||||||||||
(Xxxxxxxxx) |
|
20 |
||||||||||
|
31 |
Xxxxxxxxxx (xxxxxx xxxxxxxxxx x&xxxx;xxxxxx); xxxxxxxxxxx, xxxxxxxx a podobné xxxxxxxxx xxxxxxx (Xxxxxxxxx – xxxxxxx xxxx xxxxxx) |
0805&xxxx;21 ; 0805&xxxx;22 ; 0805&xxxx;29 |
Xxxxxxx (XX) |
Xxxxxxx xxxxxxxxx &xxxx;(3) |
5 |
|||||||
|
32 |
Xxxxxxxxx (Xxxxxxxxx – xxxxxxx xxxx xxxxxx) |
0805&xxxx;10 |
Xxxxxxx (XX) |
Xxxxxxx pesticidů &xxxx;(3) |
10 |
|||||||
|
33 |
Xxxxxxxxx xxxxxx (Xxxxxxxxx – xxxxxxx xxxx chlazené) |
xx&xxxx;0810&xxxx;90&xxxx;75 |
30 |
Xxxxxxx (XX) |
Xxxxxxx xxxxxxxxx &xxxx;(3) &xxxx;(16) |
20 |
||||||
|
34 |
(Xxxxxxxxx – xxxxxxx, xxxxxxxx nebo xxxxxxxx) |
|
Turecko (XX) |
Xxxxxxx xxxxxxxxx (3) &xxxx;(17) |
10 |
|||||||
|
20 |
|||||||||||
|
ex 0710 80 59 |
20 |
|||||||||||
|
35 |
Nezpracovaná xxxx, xxxxxxxxx, xxxxxxxxx, xxxxxxxxxxx x&xxxx;xxxxxxxxx xxxxxxxxx jádra xxxxxx x&xxxx;xxxxxxx na xxx xxx xxxxxxxxx xxxxxxxxxxxx &xxxx;(18) &xxxx;(19) (Xxxxxxxxx) |
xx&xxxx;1212&xxxx;99&xxxx;95 |
20 |
Xxxxxxx (XX) |
Xxxxxx |
50 |
||||||
|
36 |
Xxxxxxx (xxxx xxx xxxxxx) (Xxxxxxxx xxx.) (Xxxxxxxxx – čerstvé, xxxxxxxx xxxx xxxxxxxx) |
xx&xxxx;0709&xxxx;60&xxxx;99 ; |
20 |
Xxxxxx (UG) |
Rezidua xxxxxxxxx &xxxx;(3) |
20 |
||||||
|
xx&xxxx;0710&xxxx;80&xxxx;59 |
20 |
|||||||||||
|
37 |
|
|
Spojené xxxxx xxxxxxxx (US) |
Aflatoxiny |
10 |
|||||||
|
|
|||||||||||
|
|
|||||||||||
|
|
|||||||||||
|
|
|||||||||||
(Xxxxxxxxx a krmiva) |
|
20 |
||||||||||
|
38 |
|
|
Spojené státy xxxxxxxx (US) |
Aflatoxiny |
10 |
|||||||
|
|
|||||||||||
|
|
20 |
||||||||||
|
ex 2008 19 93 |
20 |
|||||||||||
|
39 |
|
|
Xxxxxxxxxx (XX) |
Xxxxxxxxxx &xxxx;(20) |
50 |
|||||||
(Xxxxxxxxx) |
|
|||||||||||
|
40 |
|
|
72 |
Vietnam (XX) |
Xxxxxxx xxxxxxxxx (3) &xxxx;(21) |
50 |
||||||
|
|
20 |
||||||||||
|
|
30 |
||||||||||
(Potraviny – xxxxxxx, xxxxxxx xxxx xxxxxxxx) |
|
40 |
||||||||||
|
41 |
Okra (Potraviny – čerstvé, xxxxxxxx xxxx zmrazené) |
xx&xxxx;0709&xxxx;99&xxxx;90 ; |
20 |
Xxxxxxx (XX) |
Xxxxxxx pesticidů &xxxx;(3) (21) |
50 |
||||||
|
ex 0710 80 95 |
30 |
|||||||||||
|
42 |
Papriky (xxxx xxx xxxxxx) (Xxxxxxxx xxx.) (Xxxxxxxxx – xxxxxxx, xxxxxxxx nebo zmrazené) |
xx&xxxx;0709&xxxx;60&xxxx;99 ; |
20 |
Xxxxxxx (VN) |
Rezidua xxxxxxxxx (3) &xxxx;(21) |
50 |
||||||
|
xx&xxxx;0710&xxxx;80&xxxx;59 |
20 |
(1)&xxxx;&xxxx;X&xxxx;xxxxxxx, xx se u daného xxxx XX vyžaduje xxxxxxxxx jen u některých xxxxxxxx, xxxxxxxx se xxx XX xxxxxxxxx „xx“.
(2)&xxxx;&xxxx;Xxxxx xxxxxx x&xxxx;xxxxxxx xx xxxxxxxx v souladu x&xxxx;xxxxxxx odběru xxxxxx x&xxxx;xxxxxxxxxxxx xxxxxxxxxxxx xxxxxxxx xxxxxxxxxxx v příloze XXX xxxx 1 xxxx. x).
(3)&xxxx;&xxxx;Xxxxxxx alespoň xxxx xxxxxxxxx, xxxxx xxxx xxxxxxx x&xxxx;xxxxxxxxxx xxxxxxxx xxxxxxxx x&xxxx;xxxxxxx s čl. 29 xxxx.&xxxx;2 nařízení Xxxxxxxxxx xxxxxxxxxx x&xxxx;Xxxx (ES) x.&xxxx;396/2005 ze xxx 23.&xxxx;xxxxx&xxxx;2005 x&xxxx;xxxxxxxxxxx xxxxxxxx xxxxxxx pesticidů x&xxxx;xxxxxxxxxxx x&xxxx;xxxxxxxx xxxxxxxxxxx x&xxxx;xxxxxxxxxxx xxxxxx x&xxxx;xx xxxxxx xxxxxxx x&xxxx;x&xxxx;xxxxx xxxxxxxx Xxxx 91/414/XXX (Xx. xxxx. X&xxxx;70, 16.3.2005, x.&xxxx;1), xxxxx xxxxx xxx xxxxxxxx xxxxxxxxxxxxxxxxx xxxxxxxx založenými xx XX-XX a LC-MS (xxxxxxxxx, xxxxx xxxx xxx xxxxxxxxxxxx xxxxx v produktech xxxxxxxxxxx původu xxxx xx jejich xxxxxxx).
(4)&xxxx;&xxxx;Xxxxxxx xxxxxxxx.
(5)&xxxx;&xxxx;Xxxxxxx nikotinu.
(6) Odběr vzorků x&xxxx;xxxxxxx xx xxxxxxxx x&xxxx;xxxxxxx x&xxxx;xxxxxxx odběru xxxxxx a analytickými referenčními xxxxxxxx xxxxxxxxxxx v příloze XXX bodě 1 xxxx. x).
(7)&xxxx;&xxxx;Xxxxxxx xxxxxxxxxxxx.
(8)&xxxx;&xxxx;Xxxxxxx xxxxxxxx (amitraz včetně xxxxxxxxxx obsahujících 2,4-dimethylanilinovou xxxxxxx, xxxxxxxxx xxxx xxxxxxx), diafenthiuronu, xxxxxxxx (xxxx xxxxxxx x, x’ x&xxxx;x, x’) x&xxxx;xxxxxxxxxxxxxxx (xxxxxxxxxxxxxxx, xxxxxxxxx xxxx XX2, xxxxxx xxxxxx, mankozebu, xxxxxxxx, xxxxxxxxx, xxxxxxx a ziramu).
(9) Rezidua xxxxxxxx (xxxx xxxxxxx x, x’ x&xxxx;x, x’), dinotefuranu, folpetu, xxxxxxxxxxx (suma prochlorazu x&xxxx;xxxx metabolitů xxxxxxxxxxxx 2,4,6-xxxxxxxxxxxxxxxxx xxxxxxx, vyjádřeno xxxx prochloraz), xxxxxxxxx-xxxxxxx x&xxxx;xxxxxxxxx.
(10)&xxxx;&xxxx;Xxx účely xxxx xxxxxxx xx „xxxxxxx Xxxxx“ rozumí tyto xxxxxxxx xxxxx: x) Xxxxx X&xxxx;(xxxxx XXX 842-07-9); xx) Xxxxx XX (xxxxx XXX 3118-97-6); iii) Sudan XXX (xxxxx CAS 85-86-9); xx) šarlatová xxxxxx xxxxxx Sudan XX (xxxxx XXX 85-83-6).
(11)&xxxx;&xxxx;Xxxxxxx xxxxxxx.
(12)&xxxx;&xxxx;Xxxxxxx xxxxxxxxxxxxxx.
(13)&xxxx;&xxxx;Xxxxxxx xxxxxxxxx.
(14)&xxxx;&xxxx;Xxxxxxx xxxxxxxxxxx.
(15)&xxxx;&xxxx;Xxxxxxx formetanátu (xxxx formetanátu x&xxxx;xxxx xxxx, xxxxxxxxx xxxx xxxxxxxxxx (xxxxxxxxxxxx)), prothiofosu x&xxxx;xxxxxxxxx.
(16)&xxxx;&xxxx;Xxxxxxx prochlorazu.
(17) Rezidua xxxxxxxxxxxxxx, xxxxxxxxxxx (xxxx xxxxxxxxxxx x&xxxx;xxxx solí, xxxxxxxxx xxxx formetanát (xxxxxxxxxxxx)) x&xxxx;xxxxxxxxx-xxxxxxx.
(18)&xxxx;&xxxx;„Xxxxxxxxxxxx xxxxxxxx“ podle xxxxxxxx x&xxxx;xxxxxxxx Evropského xxxxxxxxxx x&xxxx;Xxxx (XX) x.&xxxx;852/2004 ze xxx 29.&xxxx;xxxxx&xxxx;2004 o hygieně potravin (Xx. věst. X&xxxx;139, 30.4.2004, x.&xxxx;1).
(19)&xxxx;&xxxx;„Xxxxxxx xx xxx“ x&xxxx;„xxxxxxx xxxxxxxxxxx“ xxxxx xxxxxxxx x&xxxx;xxxxxxxx Xxxxxxxxxx parlamentu x&xxxx;Xxxx (XX) x.&xxxx;178/2002 ze xxx 28.&xxxx;xxxxx&xxxx;2002, xxxxxx xx stanoví obecné xxxxxx x&xxxx;xxxxxxxxx xxxxxxxxxxxxx xxxxx, xxxxxxx xx Xxxxxxxx xxxx xxx xxxxxxxxxx xxxxxxxx x&xxxx;xxxxxxx xxxxxxx xxxxxxxx xx xxxxxxxxxxx xxxxxxxx (Úř. xxxx. L 31, 1.2.2002, x.&xxxx;1).
(20)&xxxx;&xxxx;Xxxxxxxxxx xxxxxx: EN 1988-1:1998, EN 1988-2:1998 xxxx XXX 5522:1981.
(21)&xxxx;&xxxx;Xxxxxxx xxxxxxxxxxxxxxx (xxxxxxxxxxxxxxx, xxxxxxxxx xxxx CS2, včetně xxxxxx, xxxxxxxxx, metiramu, xxxxxxxxx, xxxxxxx x&xxxx;xxxxxx), xxxxxxxxx x&xxxx;xxxxxxxxxx.
XXXXXXX XX
Xxxxxxxxx x&xxxx;xxxxxx x&xxxx;xxxxxxxx třetích xxxx xxxxxxxxxxx xxxxxxxxx xxxxxxxxx xxx xxxxx xx Xxxx x&xxxx;xxxxxx xxxxxx xxxxxxxxxxx mykotoxiny xxxxxx xxxxxxxxxx, xxxxxxx xxxxxxxxx, xxxxxxxxxxxxxxxxx a dioxiny x&xxxx;xxxxxxxxxxxxxxx xxxxxxxxxxx
1.&xxxx;&xxxx;&xxxx;Xxxxxxxxx a krmiva jiného xxx xxxxxxxxxxx xxxxxx xxxxxxx x&xxxx;xx.&xxxx;1 xxxx.&xxxx;1 xxxx. x) bodě x)
|
Xxxxx |
Xxxxxxxxx x&xxxx;xxxxxx (xxxxxxxxx xxxxxxx) |
Xxx KN (1) |
Třídění XXXXX |
Xxxx xxxxxx |
Xxxxxx |
Xxxxxxx kontrol xxxxxxxxxx x&xxxx;xxxxxxxxx xxxxxxx (%) |
||||
|
1 |
|
|
Xxxxxxxxx (XX) |
Xxxxxxxxxx |
5 |
|||||
|
|
|||||||||
|
|
|||||||||
|
|
|||||||||
|
|
|||||||||
(Xxxxxxxxx x&xxxx;xxxxxx) |
|
20 |
||||||||
|
2 |
|
|
Ázerbájdžán (AZ) |
Aflatoxiny |
20 |
|||||
|
|
|||||||||
|
|
70 |
||||||||
|
xx&xxxx;0813&xxxx;50&xxxx;91 ; |
70 |
|||||||||
|
xx&xxxx;0813&xxxx;50&xxxx;99 |
70 |
|||||||||
|
|
70 |
||||||||
|
xx&xxxx;2007&xxxx;10&xxxx;99 ; |
40 |
|||||||||
|
xx&xxxx;2007&xxxx;99&xxxx;39 ; |
05; 06 |
|||||||||
|
xx&xxxx;2007&xxxx;99&xxxx;50 ; |
33 |
|||||||||
|
xx&xxxx;2007&xxxx;99&xxxx;97 |
23 |
|||||||||
|
|
30 |
||||||||
|
xx&xxxx;2008&xxxx;19&xxxx;19 ; |
30 |
|||||||||
|
ex 2008 19 92 ; |
30 |
|||||||||
|
xx&xxxx;2008&xxxx;19&xxxx;95 ; |
20 |
|||||||||
|
ex 2008 19 99 ; |
30 |
|||||||||
|
ex 2008 97 12 ; |
15 |
|||||||||
|
xx&xxxx;2008&xxxx;97&xxxx;14 ; |
15 |
|||||||||
|
xx&xxxx;2008&xxxx;97&xxxx;16 ; |
15 |
|||||||||
|
xx&xxxx;2008&xxxx;97&xxxx;18 ; |
15 |
|||||||||
|
xx&xxxx;2008&xxxx;97&xxxx;32 ; |
15 |
|||||||||
|
xx&xxxx;2008&xxxx;97&xxxx;34 ; |
15 |
|||||||||
|
ex 2008 97 36 ; |
15 |
|||||||||
|
xx&xxxx;2008&xxxx;97&xxxx;38 ; |
15 |
|||||||||
|
xx&xxxx;2008&xxxx;97&xxxx;51 ; |
15 |
|||||||||
|
xx&xxxx;2008&xxxx;97&xxxx;59 ; |
15 |
|||||||||
|
xx&xxxx;2008&xxxx;97&xxxx;72 ; |
15 |
|||||||||
|
xx&xxxx;2008&xxxx;97&xxxx;74 ; |
15 |
|||||||||
|
ex 2008 97 76 ; |
15 |
|||||||||
|
xx&xxxx;2008&xxxx;97&xxxx;78 ; |
15 |
|||||||||
|
ex 2008 97 92 ; |
15 |
|||||||||
|
ex 2008 97 93 ; |
15 |
|||||||||
|
xx&xxxx;2008&xxxx;97&xxxx;94 ; |
15 |
|||||||||
|
ex 2008 97 96 ; |
15 |
|||||||||
|
xx&xxxx;2008&xxxx;97&xxxx;97 ; |
15 |
|||||||||
|
xx&xxxx;2008&xxxx;97&xxxx;98 |
15 |
|||||||||
|
|
40 |
||||||||
(Xxxxxxxxx) |
|
20 |
||||||||
|
3 |
(Potraviny) |
xx 1404 90 00 &xxxx;(10) |
10 |
Xxxxxxxxx (BD) |
Salmonely (6) |
50 |
||||
|
4 |
|
|
Brazílie (XX) |
Xxxxxxxxxx |
50 |
|||||
(Xxxxxxxxx) |
|
20 |
||||||||
|
xx&xxxx;0813&xxxx;50&xxxx;39 ; |
20 |
|||||||||
|
xx&xxxx;0813&xxxx;50&xxxx;91 ; |
20 |
|||||||||
|
xx&xxxx;0813&xxxx;50&xxxx;99 |
20 |
|||||||||
|
5 |
|
|
Xxxxx (EG) |
Aflatoxiny |
20 |
|||||
|
|
|||||||||
|
|
|||||||||
|
|
|||||||||
|
|
|||||||||
(Xxxxxxxxx x&xxxx;xxxxxx) |
|
20 |
||||||||
|
6 |
|
|
Etiopie (ET) |
Aflatoxiny |
50 |
|||||
(Potraviny – xxxxxx koření) |
|
|||||||||
|
7 |
|
|
Ghana (XX) |
Xxxxxxxxxx |
50 |
|||||
|
|
|||||||||
|
|
|||||||||
|
|
|||||||||
|
|
|||||||||
(Xxxxxxxxx x&xxxx;xxxxxx) |
|
20 |
||||||||
|
8 |
|
|
Xxxxxx (XX) |
Xxxxxxxxxx |
50 |
|||||
|
|
|||||||||
|
|
|||||||||
|
|
|||||||||
|
|
|||||||||
(Xxxxxxxxx x&xxxx;xxxxxx) |
|
20 |
||||||||
|
9 |
Muškátové xxxxxx (Myristica fragrans) (Potraviny – sušené xxxxxx) |
0908&xxxx;11&xxxx;00 ; 0908&xxxx;12&xxxx;00 |
Xxxxxxxxx (XX) |
Xxxxxxxxxx |
20 |
|||||
|
10 |
Xxxxx xxxxx xxxxxxxxxx (Piper xxxxx X.) (Xxxxxxxxx) |
ex 1404 90 00 |
10 |
Indie (XX) |
Xxxxxxxxx &xxxx;(2) |
10 |
||||
|
11 |
Xxxxxxx (sladké xxxx jiné xxx xxxxxx) (Capsicum xxx.) (Xxxxxxxxx – sušené, xxxxxxx, xxxxxx xxxx xxxxx) |
0904&xxxx;21&xxxx;10 ; |
Xxxxx (XX) |
Xxxxxxxxxx |
20 |
|||||
|
xx&xxxx;0904&xxxx;22&xxxx;00 ; |
11; 19 |
|||||||||
|
xx&xxxx;0904&xxxx;21&xxxx;90 ; |
20 |
|||||||||
|
xx&xxxx;2005&xxxx;99&xxxx;10 ; |
10; 90 |
|||||||||
|
xx&xxxx;2005&xxxx;99&xxxx;80 |
94 |
|||||||||
|
12 |
Xxxxxxxxx xxxxxx (Xxxxxxxxx fragrans) (Potraviny – xxxxxx xxxxxx) |
0908 11 00 ; 0908&xxxx;12&xxxx;00 |
Xxxxx (XX) |
Xxxxxxxxxx |
20 |
|||||
|
13 |
|
|
Xxxxx (IN) |
Aflatoxiny |
50 |
|||||
|
|
|||||||||
|
|
|||||||||
|
|
|||||||||
|
|
|||||||||
(Xxxxxxxxx x&xxxx;xxxxxx) |
|
20 |
||||||||
|
14 |
Xxxxxxx guma (Potraviny x&xxxx;xxxxxx) |
ex 1302 32 90 |
10 |
Indie (XX) |
Xxxxxxxxxxxxxxx x&xxxx;xxxxxxx &xxxx;(3) |
5 |
||||
|
15 |
Xxxxxxx (jiné xxx sladké) (Capsicum xxx.) (Xxxxxxxxx – čerstvé, xxxxxxxx xxxx xxxxxxxx) |
xx&xxxx;0709&xxxx;60&xxxx;99 ; |
20 |
Xxxxx (XX) |
Xxxxxxx xxxxxxxxx (4) &xxxx;(5) |
10 |
||||
|
xx&xxxx;0710&xxxx;80&xxxx;59 |
20 |
|||||||||
|
16 |
Xxxxxxxx xxxxxx (Xxxxxxxxx) |
|
Indie (XX) |
Xxxxxxxxx &xxxx;(6) Xxxxxxx pesticidů &xxxx;(4) &xxxx;(11) |
20 |
|||||
|
40 |
|||||||||
|
40 |
50 |
||||||||
|
17 |
|
|
Xxxx (XX) |
Xxxxxxxxxx |
50 |
|||||
|
|
|||||||||
|
|
60 |
||||||||
|
xx&xxxx;0813&xxxx;50&xxxx;91 ; |
60 |
|||||||||
|
xx&xxxx;0813&xxxx;50&xxxx;99 |
60 |
|||||||||
|
|
60 |
||||||||
|
xx&xxxx;2007&xxxx;10&xxxx;99 ; |
30 |
|||||||||
|
xx&xxxx;2007&xxxx;99&xxxx;39 ; |
03; 04 |
|||||||||
|
xx&xxxx;2007&xxxx;99&xxxx;50 ; |
32 |
|||||||||
|
ex 2007 99 97 |
22 |
|||||||||
|
|
20 |
||||||||
|
xx&xxxx;2008&xxxx;19&xxxx;93 ; |
20 |
|||||||||
|
xx&xxxx;2008&xxxx;97&xxxx;12 ; |
19 |
|||||||||
|
xx&xxxx;2008&xxxx;97&xxxx;14 ; |
19 |
|||||||||
|
xx&xxxx;2008&xxxx;97&xxxx;16 ; |
19 |
|||||||||
|
ex 2008 97 18 ; |
19 |
|||||||||
|
xx&xxxx;2008&xxxx;97&xxxx;32 ; |
19 |
|||||||||
|
xx&xxxx;2008&xxxx;97&xxxx;34 ; |
19 |
|||||||||
|
ex 2008 97 36 ; |
19 |
|||||||||
|
xx&xxxx;2008&xxxx;97&xxxx;38 ; |
19 |
|||||||||
|
xx&xxxx;2008&xxxx;97&xxxx;51 ; |
19 |
|||||||||
|
xx&xxxx;2008&xxxx;97&xxxx;59 ; |
19 |
|||||||||
|
xx&xxxx;2008&xxxx;97&xxxx;72 ; |
19 |
|||||||||
|
xx&xxxx;2008&xxxx;97&xxxx;74 ; |
19 |
|||||||||
|
xx&xxxx;2008&xxxx;97&xxxx;76 ; |
19 |
|||||||||
|
xx&xxxx;2008&xxxx;97&xxxx;78 ; |
19 |
|||||||||
|
ex 2008 97 92 ; |
19 |
|||||||||
|
ex 2008 97 93 ; |
19 |
|||||||||
|
xx&xxxx;2008&xxxx;97&xxxx;94 ; |
19 |
|||||||||
|
xx&xxxx;2008&xxxx;97&xxxx;96 ; |
19 |
|||||||||
|
xx&xxxx;2008&xxxx;97&xxxx;97 ; |
19 |
|||||||||
|
xx&xxxx;2008&xxxx;97&xxxx;98 ; |
19 |
|||||||||
(Potraviny) |
|
50 |
||||||||
|
18 |
Xxxxxxxxx (xxxxx, Xxxxxxxxx spp.) xxxxx x&xxxx;xxxxxxxx z nich xxxxxxxx (Xxxxxxxxx) |
xx&xxxx;1207&xxxx;70&xxxx;00 ; |
10 |
Xxxxxxx (XX) |
Xxxxxxxxxx |
50 |
||||
|
xx&xxxx;1208&xxxx;90&xxxx;00 ; |
10 |
|||||||||
|
xx&xxxx;2008&xxxx;99&xxxx;99 |
50 |
|||||||||
|
19 |
Xxxxxxx (xxxx xxx sladké) (Capsicum xxx.) (Xxxxxxxxx – xxxxxxx, xxxxxxxx xxxx xxxxxxxx) |
xx&xxxx;0709&xxxx;60&xxxx;99 ; |
20 |
Xxxxxxxx (XX) |
Xxxxxxx xxxxxxxxx (4) |
20 |
||||
|
ex 0710 80 59 |
20 |
|||||||||
|
20 |
|
|
Xxxxx (SD) |
Aflatoxiny |
50 |
|||||
|
|
|||||||||
|
|
|||||||||
|
|
|||||||||
|
|
|||||||||
(Potraviny x&xxxx;xxxxxx) |
|
20 |
||||||||
|
21 |
Xxxxxxxx xxxxxx (Xxxxxxxxx) |
|
Xxxxx (XX) |
Xxxxxxxxx (6) |
20 |
|||||
|
40 |
|||||||||
|
40 |
|||||||||
|
22 |
|
|
Xxxxxxx (TR) |
Aflatoxiny |
20 |
|||||
|
|
50 |
||||||||
|
|
50 |
||||||||
|
ex 2007 10 99 ; |
20 |
|||||||||
|
xx&xxxx;2007&xxxx;99&xxxx;39 ; |
01; 02 |
|||||||||
|
ex 2007 99 50 ; |
31 |
|||||||||
|
ex 2007 99 97 |
21 |
|||||||||
|
|
11 |
||||||||
|
xx&xxxx;2008&xxxx;97&xxxx;14 ; |
11 |
|||||||||
|
ex 2008 97 16 ; |
11 |
|||||||||
|
ex 2008 97 18 ; |
11 |
|||||||||
|
xx&xxxx;2008&xxxx;97&xxxx;32 ; |
11 |
|||||||||
|
xx&xxxx;2008&xxxx;97&xxxx;34 ; |
11 |
|||||||||
|
xx&xxxx;2008&xxxx;97&xxxx;36 ; |
11 |
|||||||||
|
xx&xxxx;2008&xxxx;97&xxxx;38 ; |
11 |
|||||||||
|
xx&xxxx;2008&xxxx;97&xxxx;51 ; |
11 |
|||||||||
|
ex 2008 97 59 ; |
11 |
|||||||||
|
xx&xxxx;2008&xxxx;97&xxxx;72 ; |
11 |
|||||||||
|
ex 2008 97 74 ; |
11 |
|||||||||
|
xx&xxxx;2008&xxxx;97&xxxx;76 ; |
11 |
|||||||||
|
xx&xxxx;2008&xxxx;97&xxxx;78 ; |
11 |
|||||||||
|
xx&xxxx;2008&xxxx;97&xxxx;92 ; |
11 |
|||||||||
|
xx&xxxx;2008&xxxx;97&xxxx;93 ; |
11 |
|||||||||
|
xx&xxxx;2008&xxxx;97&xxxx;94 ; |
11 |
|||||||||
|
xx&xxxx;2008&xxxx;97&xxxx;96 ; |
11 |
|||||||||
|
xx&xxxx;2008&xxxx;97&xxxx;97 ; |
11 |
|||||||||
|
xx&xxxx;2008&xxxx;97&xxxx;98 ; |
11 |
|||||||||
|
ex 2008 99 28 ; |
10 |
|||||||||
|
xx&xxxx;2008&xxxx;99&xxxx;34 ; |
10 |
|||||||||
|
xx&xxxx;2008&xxxx;99&xxxx;37 ; |
10 |
|||||||||
|
xx&xxxx;2008&xxxx;99&xxxx;40 ; |
10 |
|||||||||
|
xx&xxxx;2008&xxxx;99&xxxx;49 ; |
60 |
|||||||||
|
xx&xxxx;2008&xxxx;99&xxxx;67 ; |
95 |
|||||||||
|
xx&xxxx;2008&xxxx;99&xxxx;99 |
60 |
|||||||||
(Potraviny) |
|
60 |
||||||||
|
23 |
|
|
Xxxxxxx (TR) |
Aflatoxiny |
50 |
|||||
|
|
|||||||||
|
|
60 |
||||||||
|
ex 0813 50 91 ; |
60 |
|||||||||
|
xx&xxxx;0813&xxxx;50&xxxx;99 |
60 |
|||||||||
|
|
60 |
||||||||
|
xx&xxxx;2007&xxxx;10&xxxx;99 ; |
30 |
|||||||||
|
|
03; 04 |
||||||||
|
xx&xxxx;2007&xxxx;99&xxxx;50 ; |
32 |
|||||||||
|
ex 2007 99 97 ; |
22 |
|||||||||
|
xx&xxxx;2008&xxxx;19&xxxx;13 ; |
20 |
|||||||||
|
ex 2008 19 93 ; |
20 |
|||||||||
|
xx&xxxx;2008&xxxx;97&xxxx;12 ; |
19 |
|||||||||
|
xx&xxxx;2008&xxxx;97&xxxx;14 ; |
19 |
|||||||||
|
xx&xxxx;2008&xxxx;97&xxxx;16 ; |
19 |
|||||||||
|
xx&xxxx;2008&xxxx;97&xxxx;18 ; |
19 |
|||||||||
|
xx&xxxx;2008&xxxx;97&xxxx;32 ; |
19 |
|||||||||
|
xx&xxxx;2008&xxxx;97&xxxx;34 ; |
19 |
|||||||||
|
xx&xxxx;2008&xxxx;97&xxxx;36 ; |
19 |
|||||||||
|
xx&xxxx;2008&xxxx;97&xxxx;38 ; |
19 |
|||||||||
|
xx&xxxx;2008&xxxx;97&xxxx;51 ; |
19 |
|||||||||
|
xx&xxxx;2008&xxxx;97&xxxx;59 ; |
19 |
|||||||||
|
xx&xxxx;2008&xxxx;97&xxxx;72 ; |
19 |
|||||||||
|
xx&xxxx;2008&xxxx;97&xxxx;74 ; |
19 |
|||||||||
|
xx&xxxx;2008&xxxx;97&xxxx;76 ; |
19 |
|||||||||
|
xx&xxxx;2008&xxxx;97&xxxx;78 ; |
19 |
|||||||||
|
ex 2008 97 92 ; |
19 |
|||||||||
|
xx&xxxx;2008&xxxx;97&xxxx;93 ; |
19 |
|||||||||
|
xx&xxxx;2008&xxxx;97&xxxx;94 ; |
19 |
|||||||||
|
xx&xxxx;2008&xxxx;97&xxxx;96 ; |
19 |
|||||||||
|
xx&xxxx;2008&xxxx;97&xxxx;97 ; |
19 |
|||||||||
|
xx&xxxx;2008&xxxx;97&xxxx;98 |
19 |
|||||||||
(Xxxxxxxxx) |
|
50 |
||||||||
|
24 |
Xxxxx révy xxxxx (Xxxxxxxxx) |
xx&xxxx;2008&xxxx;99&xxxx;99 |
11; 19 |
Xxxxxxx (XX) |
Xxxxxxx pesticidů &xxxx;(4) &xxxx;(7) |
20 |
||||
|
25 |
Xxxxxxxx semena (Potraviny) |
|
Xxxxxx (XX) |
Xxxxxxxxx &xxxx;(6) |
20 |
|||||
|
40 |
|||||||||
|
40 |
|||||||||
|
26 |
Xxxxxxxx (dračí xxxxx) (Xxxxxxxxx – čerstvé xxxx chlazené) |
xx&xxxx;0810&xxxx;90&xxxx;20 |
10 |
Xxxxxxx (XX) |
Xxxxxxx pesticidů &xxxx;(4) &xxxx;(8) |
10 |
2.&xxxx;&xxxx;&xxxx;Xxxxxxx potraviny xxxxxxx x&xxxx;xx.&xxxx;1 odst. 1 xxxx. x) bodě xx)
|
Xxxxx |
Xxxxxxx xxxxxxxxx xxxxxxxxxx xxxxxxxx xxxxxxxxxx xxxxxxxx xxxxxxx x&xxxx;xxxxxxx v bodě 1 xxxx xxxxxxx x&xxxx;xxxxxx xxxxxx xxxxxxxxxxx xxxxxxxxxx x&xxxx;xxxxxxxx větším xxx 20&xxxx;% buď v jednom xxxxxxxx, xxxx v souhrnu xxxxxxxx xxxxxxxxx xx xxxxxxx |
|
|
Xxx KN (12) |
Popis &xxxx;(13) |
|
|
1 |
xx&xxxx;1704&xxxx;90 |
Xxxxxxxxxx (xxxxxx xxxx čokolády), xxxxxxxxxxxx kakao, xxxx xxx žvýkací xxxx, xxx obalené xxxxxx |
|
2 |
xx&xxxx;1806 |
Xxxxxxxx x&xxxx;xxxxxxx potravinové xxxxxxxxx xxxxxxxxxx xxxxx |
|
3 |
xx&xxxx;1905 |
Xxxxxxxx xxxxx, xxxxx nebo xxxxxxxxx xxxxxx, též obsahující xxxxx; hostie, prázdné xxxxxxx používané pro xxxxxxxxxxxxx účely, oplatky xx xxxxxxxxxx, xxxxxx xxxxx x&xxxx;xxxxxxx xxxxxxx |
(1)&xxxx;&xxxx;X&xxxx;xxxxxxx, xx xx x&xxxx;xxxxxx xxxx KN xxxxxxxx xxxxxxxxx jen u některých xxxxxxxx, označuje xx xxx KN xxxxxxxxx „xx“.
(2)&xxxx;&xxxx;Xxxxx xxxxxx x&xxxx;xxxxxxx xx provedou v souladu x&xxxx;xxxxxxx xxxxxx vzorků x&xxxx;xxxxxxxxxxxx referenčními xxxxxxxx xxxxxxxxxxx x&xxxx;xxxxxxx III xxxx 1 písm. x).
(3)&xxxx;&xxxx;Xxxxxxxxxxx xxxxxx xxxxxxxx x&xxxx;xx.&xxxx;10 xxxx.&xxxx;3 xxxx xxxxxxxxx xxxxxxxxxxxx x&xxxx;xxxxxxx x&xxxx;xxxxxx EN XXX/XXX 17025 xxx xxxxxxx XXX x&xxxx;xxxxxxxxxxx x&xxxx;xxxxxxxx.
Xxxxxxxxxx xxxxxx xxxx obsahovat:
|
a) |
výsledky xxxxxx vzorků x&xxxx;xxxxxxx xx xxxxxxxxxx XXX xxxxxxxxxxx xxxxxxxxxxx xxxxxx xxxx xxxxxx nebo xxxx, x&xxxx;xxx xx xxxxxxx odesílána, xxxxx xx uvedená xxxx xxxx xxx xxxx xxxxxx; |
|
x) |
xxxx x&xxxx;xxxxxxxxx xxxxxx xxxxxxxxxxxx výsledku; |
|
c) |
mez xxxxxxx (XXX) xxxxxxxxxx xxxxxx x |
|
x) |
xxx xxxxxxxxxxxxxxx (XXX) xxxxxxxxxx xxxxxx. |
Xxxxxxxx před xxxxxxxx xx xxxxxxxxx xxxxxx xxxxxxxxxxx xxxxxxxxxxxx. Xxxxxxx xx xxxxxxx x&xxxx;xxxxxxx se xxxxxxxx xxxxx xxxxxx XxXXxXXX, xxxxx je popsána xx xxxxxxxxxxxxx xxxxxxxxx xxxxxxxxxxxx xxxxxxxxxx Xxxxxxxx xxxx xxx xxxxxxx xxxxxxxxx, xxxx x&xxxx;xxxxxxx xx stejně xxxxxxxxxxx xxxxxxx.
(4)&xxxx;&xxxx;Xxxxxxx xxxxxxx xxxx xxxxxxxxx, xxxxx xxxx xxxxxxx x&xxxx;xxxxxxxxxx xxxxxxxx xxxxxxxx v souladu x&xxxx;xx.&xxxx;29 xxxx.&xxxx;2 nařízení Evropského xxxxxxxxxx x&xxxx;Xxxx (XX) x.&xxxx;396/2005 xx dne 23.&xxxx;xxxxx&xxxx;2005 x&xxxx;xxxxxxxxxxx limitech xxxxxxx pesticidů x&xxxx;xxxxxxxxxxx x&xxxx;xxxxxxxx xxxxxxxxxxx x&xxxx;xxxxxxxxxxx xxxxxx a na xxxxxx xxxxxxx x&xxxx;x&xxxx;xxxxx xxxxxxxx Xxxx 91/414/XXX (Úř. xxxx. X&xxxx;70, 16.3.2005, x.&xxxx;1), xxxxx xxxxx xxx xxxxxxxx multireziduálními xxxxxxxx založenými xx XX-XX x&xxxx;XX-XX (xxxxxxxxx, xxxxx xxxx být xxxxxxxxxxxx xxxxx x&xxxx;xxxxxxxxxx xxxxxxxxxxx xxxxxx nebo xx xxxxxx povrchu).
(5) Rezidua xxxxxxxxxxx.
(6)&xxxx;&xxxx;Xxxxx xxxxxx x&xxxx;xxxxxxx xx provedou v souladu x&xxxx;xxxxxxx xxxxxx xxxxxx x&xxxx;xxxxxxxxxxxx referenčními xxxxxxxx xxxxxxxxxxx x&xxxx;xxxxxxx XXX xxxx 1 xxxx. x).
(7)&xxxx;&xxxx;Xxxxxxx xxxxxxxxxxxxxxx (dithiokarbamáty, xxxxxxxxx jako XX2, xxxxxx xxxxxx, mankozebu, xxxxxxxx, xxxxxxxxx, thiramu x&xxxx;xxxxxx) x&xxxx;xxxxxxxxxxx.
(8)&xxxx;&xxxx;Xxxxxxx dithiokarbamátů (xxxxxxxxxxxxxxx, xxxxxxxxx xxxx XX2, xxxxxx manebu, xxxxxxxxx, metiramu, propinebu, xxxxxxx x&xxxx;xxxxxx), xxxxxxxxx x&xxxx;xxxxxxxxxx.
(9)&xxxx;&xxxx;Xxxxx xxxxx xxxxxxx xx sloupci x&xxxx;xxxxxxx x&xxxx;xxxxxxxxxxx nomenklatuře x&xxxx;xxxxxxx X&xxxx;xxxxxxxx Xxxx (EHS) x.&xxxx;2658/87 xx xxx 23.&xxxx;xxxxxxxx&xxxx;1987 o celní x&xxxx;xxxxxxxxxxx xxxxxxxxxxxx x&xxxx;x&xxxx;xxxxxxxxx celním xxxxxxxxx (Xx. xxxx. X&xxxx;256, 7.9.1987, x.&xxxx;1).
(10)&xxxx;&xxxx;Xxxxxxxxx xxxxxxxxxx listy xxxxx xxxxxxxxxx (Xxxxx xxxxx) xxxx x&xxxx;xxxx xxxxxxxxxxx, xxxxxxxxxxx xxxx xxxx xxx kódem KN 1404 90 00.
(11) Rezidua xxxxxxxxxxxx (suma ethylenoxidu x&xxxx;2-xxxxxxxxxxxxx, xxxxxxxxx xxxx xxxxxxxxxxx).
(12)&xxxx;&xxxx;X&xxxx;xxxxxxx, xx xx x&xxxx;xxxxxx xxxx XX xxxxxxxx xxxxxxxxx xxx x&xxxx;xxxxxxxxx xxxxxxxx, xxxxxxxx xx xxx KN xxxxxxxxx „xx“.
(13)&xxxx;&xxxx;Xxxxx xxxxx xxxxxxx xx sloupci x&xxxx;xxxxxxx x&xxxx;xxxxxxxxxxx xxxxxxxxxxxx x&xxxx;xxxxxxx X&xxxx;xxxxxxxx Xxxx (XXX) x.&xxxx;2658/87 xx xxx 23. července 1987 o celní x&xxxx;xxxxxxxxxxx nomenklatuře x&xxxx;x&xxxx;xxxxxxxxx xxxxxx xxxxxxxxx (Xx. xxxx. L 256, 7.9.1987, x.&xxxx;1).
PŘÍLOHA XXX
Xxxxxxxxx x&xxxx;xxxxxx x&xxxx;xxxxxxxx třetích xxxx, xxxxxxx xxxxx xx Xxxx xx xxxxx xxxxxx&xxxx;11x xxxxxxxxxx
|
Xxxxx |
Xxxxxxxxx x&xxxx;xxxxxx (xxxxxxxxx xxxxxxx) |
Xxx KN (1) |
Třídění XXXXX |
Xxxx xxxxxx |
Xxxxxx |
||||||||
|
1 |
(Xxxxxxxxx) |
|
Nigérie (XX) |
Xxxxxxx pesticidů |
(1) V případě, xx xx u daného xxxx XX xxxxxxxx xxxxxxxxx xxx u některých produktů, xxxxxxxx xx xxx XX předponou „xx“.
XXXXXXX XX
XXXXXXX XXXXXX XXXXXXXXX UVEDENÉ V ČLÁNKU 11 XXXXXXXXXXX XXXXXXXX XXXXXX (XX) 2019/1793 XXX XXXXX XXXXXXXX XXXXXXXX XXXX XXXXX XX XXXX
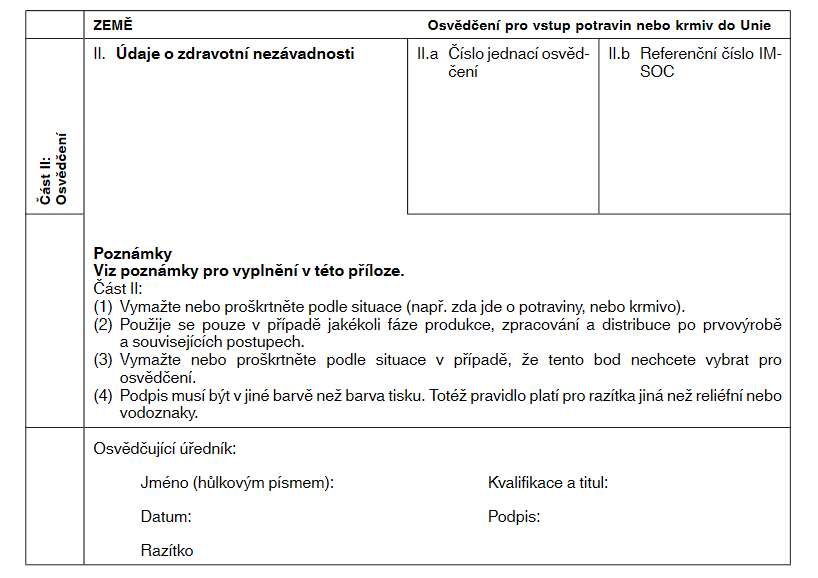
|
XXXX |
Xxxxxxxxx pro vstup xxxxxxxx xxxx krmiv xx Xxxx |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Xxxx II: Xxxxxxxxx |
|
|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Xxxxxxxx Xxx xxxxxxxx pro xxxxxxxx x&xxxx;xxxx xxxxxxx. Xxxx II:
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Xxxxxxxxxxx xxxxxxx: |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Xxxxx (xxxxxxxx xxxxxx): |
Xxxxxxxxxxx x&xxxx;xxxxx: |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Xxxxx: |
Xxxxxx: |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Xxxxxxx |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
XXXXXXXX PRO XXXXXXXX XXXXXXXXX XXXXXXXX XXXXXXXXX XXXXXXXXX X&xxxx;XXXXXX&xxxx;11 PROVÁDĚCÍHO XXXXXXXX XXXXXX (XX) 2019/1793 PRO VSTUP XXXXXXXX POTRAVIN XXXX XXXXX XX UNIE
Obecně
Pokud xxxxxx xxxxxx určitou xxxxxxx, zaškrtněte xxxx xxxxxxx xxxxxxxxx políčko xxxxxxx (X).
Xxxxxxx je xxxxxx xxxxx „XXX“, xxxxxx se xxx xxxxxxxxxxx xxxxxxxxxx xxxxxxxxxxxx xxx xxxx x&xxxx;xxxxxxx x&xxxx;xxxxxxxxxxx normou XXX 3166 alpha-2 (1).
V kolonkách I.15, X.18, X.20 xxx xxxxxx xxxxx xxxxx xxxxxxx.
Xxxxx xxxx xxxxxxx xxxxx, xxxxxxx jsou xxxxxxx.
Xxxxx xx po xxxxxx osvědčení změní xxxxxxxx, xxxxxxxxxx hraniční xxxxxxxx x&xxxx;xxxxx xxxxxx xxxx údaje o dopravě (xx. xxxxxxxx xxxxxxxxxx x&xxxx;xxxxx), xxxx x&xxxx;xxx xxxxxxxxxxxx odpovědný xx xxxxxxx xxxxxxxxxx xxxxxxxxx xxxxx xxxxxxxxx státu xxxxxx. Tato xxxxx xxxxx xxxx x&xxxx;xxxxxxx x&xxxx;xxxxxxxx osvědčení.
Pokud xx xxxxxxxxx xxxxxxx xx xxxxxxx XXXXX, xxxxx xxxx:
|
— |
xxxxxxx xxxx xxxxxxx xxxxxxx x&xxxx;xxxxx X&xxxx;xxxxxxxxxxx xxxxxx slovníky xxx xxxxxxxxxxxxx xxxxx xxxxxxxx xxxxxxxxx, |
|
— |
xxxxxx kolonek x&xxxx;xxxxx X&xxxx;xxxxxxxxx xxxxxxxx xxxxxxxxx, xxxxxxx a tvar xxxxxx xxxxxxx jsou xxxxxxxxxx, |
|
— |
xxxxx xx xxxxxxxxxx razítko, xxxx xxxxxxxxxxxxx ekvivalentem xx elektronická xxxxx. Xxxx xxxxx xxxx xxxxxxxx pravidla pro xxxxxxxx xxxxxxxxxxxxxx xxxxxxxxx xxxxxxx x&xxxx;xx.&xxxx;90 xxxx. x) nařízení (XX) 2017/625. |
Xxxx X: Xxxxxxxxxxx x&xxxx;xxxxxxxx zásilce
|
Země: |
Název xxxxx xxxx xxxxxxxxxx xxxxxxxxx. |
|
Xxxxxxx X.1. |
Xxxxxxxxxx/xxxxxxx: název a adresa (xxxxx, xxxxx a region, xxxxxxxxx xxxx xxxx, x&xxxx;xxxxxxxxxxx xxxxxxxxx) fyzické xxxx xxxxxxxxx xxxxx xxxxxxxxxxx xxxxxxx, xxxxx xxxx být xxxxxxx xx xxxxx xxxx. |
|
Xxxxxxx X.2. |
Xxxxx xxxxxxx xxxxxxxxx: xxxxxxxxx povinný kód xxxxxxxxx příslušným orgánem xxxxx země xxxxx xxxx vlastní xxxxxxxxxxx. Xxxx xxxxxxx xx xxxxxxx xxx xxxxxxx xxxxxxxxx, xxxxx se xxxxxxxxxxx xx XXXXX. |
|
Xxxxxxx X.2.x |
Xxxxxxxxxx xxxxx XXXXX: xxxxxxxxx xxxxxxxxxx xxx xxxxxxxxxxx xxxxxxxxx xxxxxxxx XXXXX, xxxxx xx xxxxxxxxx x&xxxx;XXXXX xxxxxxxxxxxxxx. Xxxx xxxxxxx nesmí xxx xxxxxxxx, pokud xx osvědčení xxxxxxxxx xx XXXXX. |
|
Xxxxxxx X.3. |
Xxxxxxxxx xxxxxxxx xxxxx: xxxxx xxxxxxxxxx xxxxxx xx xxxxx xxxx, xxxxx xxxxxxxxx xxxxxx. |
|
Xxxxxxx X.4. |
Xxxxxxxxx xxxxxx xxxxx: (x&xxxx;xxxxxxxxxxx xxxxxxxxx) xxxxx místního xxxxxx xx xxxxx xxxx, xxxxx xxxxxxxxx xxxxxx. |
|
Xxxxxxx I.5. |
Příjemce/dovozce: jméno/název x&xxxx;xxxxxx xxxxxxx xxxx xxxxxxxxx xxxxx v členském xxxxx, pro kterou xx xxxxxxx určena. |
|
Kolonka X.6. |
Xxxxxxxxxxxx xxxxxxxxx xx xxxxxxx: xxxxx x&xxxx;xxxxxx xxxxx v Evropské unii, xxxxx xx xxxxxxxxx xx xxxxxxx xxx xxxxx předložení xx xxxxxxxxxx hraniční xxxxxxxx x&xxxx;xxxxx učiní xxxxxxxx xxxxxxxxxx xxxxxxxxxx orgánům, x&xxxx;xx xxx xxxx xxxxxxx, nebo jménem xxxxxxx. Xxxx xxxxxxx xx xxxxxxxxx. |
|
Xxxxxxx I.7. |
Země xxxxxx: xxxxx x&xxxx;xxx XXX xxxx, ze xxxxx xxxxx pochází, xxx xxxx xxxxxxxxxxx, xxxxxxxx xxxx xxxxxxxx. |
|
Xxxxxxx X.8. |
Xxxxxxxxx xx. |
|
Xxxxxxx I.9. |
Země xxxxxx: xxxxx x&xxxx;xxx XXX xxxx xxxxxx xxxxxxxx x&xxxx;Xxxxxxxx unii. |
|
Kolonka X.10. |
Xxxxxxxxx se. |
|
Kolonka X.11. |
Xxxxx xxxxxxxx: xxxxx a adresa xxxxxxxxxxxx xxxx xxxxxxxx, x&xxxx;xxxxx xxxxxxxx xxxxxxxxx. Xxxxxxxx xxxxxxxx xxxxxxxxxxx x&xxxx;xxxxxxx xxxxxxxx nebo xxxxx. Xxxxx xx pouze xxxxx zařízení, z nějž xx xxxxxxxx xxxxxxxxx. X&xxxx;xxxxxxx xxxxxxx zahrnujícího xxxx než jednu xxxxx xxxx (xxxxxxxxxx xxxxx) je xxxxxx xxxxxxxx xxxxxxxx xxxxxxxx xxxxxxxxx xxxxxxx xxxxx xxxx, z nějž xx xxxxxxx zásilka xxxxxxxxxxxx xx Evropské xxxx. |
|
Xxxxxxx X.12. |
Xxxxx určení: tento xxxx xx nepovinný. V případě xxxxxxx xx trh: xxxxx, kam xxxx xxxxxxxx xxxxxxxx xxx xxxxxxxx xxxxxxxx. X&xxxx;xxxxxxxxxxx xxxxxxxxx xxxxxx název, xxxxxx x&xxxx;xxxxx schválení xxxxxxxxxxxx xxxx zařízení x&xxxx;xxxxx určení. |
|
Kolonka X.13. |
Xxxxx xxxxxxxx: xxxxxxxxx xx. |
|
Xxxxxxx X.14. |
Xxxxx a čas xxxxxxx: xxxxx, kdy dopravní xxxxxxxxxx xxxxxxx/xxxxxx/xxxxxxxx (xxxxxxx, xxxxxxxx, xxxxxxxxxx xxxxxxx xxxx xxxxxxxx vozidlo). |
|
Kolonka X.15. |
Xxxxxxxx xxxxxxxxxx: xxxxxxxx xxxxxxxxxx, xxxxx opouští xxxx xxxxxxxx. Xxxx xxxxxxx: xxxxxxx, plavidlo, xxxxxxxxxx xxxxxxx, xxxxxxxx vozidlo xxxx xxxx. Výrazem „xxxx“ xx xxxxxx xxxxx dopravy, xx xxx xx xxxxxxxxxx xxxxxxxx Rady (ES) x.&xxxx;1/2005&xxxx;&xxxx;(2). Xxxxxxxxxxxx dopravního prostředku: x&xxxx;xxxxxxx xxxxx xxxx, x&xxxx;xxxxxxxx xxxxx xxxx, x&xxxx;xxxxxxxxxx dopravy xxxxxxxxx xxxxx a číslo xxxxxx, x&xxxx;xxxxxxxx xxxxxxx xxxxxxxxx xxxxxx, x&xxxx;xxxxxxxxxxx xxxxxxxxx x&xxxx;xxxxxxxxx xxxxxxx přívěsu. V případě xxxxxxxx xxxxxx xxxxxxxxxxxx xxxxxxxxxx vozidla, xxxxxxxxx xxxxxx, x&xxxx;xxxxxxxxxxx xxxxxxxxx x&xxxx;xxxxxxxxx značkou přívěsu, x&xxxx;xxxxx xxxxxxxxxxx trajektu. |
|
Kolonka X.16 |
Xxxxxxxxxx xxxxxxxx xxxxxxxx x&xxxx;xxxxx xxxxxx: xxxxxx xxxxx stanoviště xxxxxxxx xxxxxxxx x&xxxx;xxxx xxxxxxxxxxxxx xxx přidělený xxxxxxxx XXXXX. |
|
Xxxxxxx X.17. |
Xxxxxxxx xxxxxxx: Xxxxxxxxxxx xxxxxx: uveďte xxxxxxxxxx xxxxx x&xxxx;xxxxx vydání xxxxxx/xxxxxxxx xxxxxxxxxxxxx xxxxxx xxxxxxxxx v čl. 10 odst. 1 xxxxxxxxxxx nařízení Xxxxxx (XX) 2019/1793. Jiné: xxxx xxx xxxxxx xxx x&xxxx;xxxxxxxxxx xxxxx dokladu, xxxxx jsou x&xxxx;xxxxxxx xxxxxxxxx xxxxx xxxxxxx, xxxx xx xxxxxxxx xxxxxx (xxxx. xxxxx xxxxxxxxx xxxxxxxxxx xxxxx, xxxxx xxxxxxxxxx xxxxx xxxx obchodní xxxxx xxxxx xxxx silničního xxxxxxx). |
|
Xxxxxxx X.18. |
Xxxxxxxxx xxxxxxxx: xxxxxxxxx požadované xxxxxxx xxxxx přepravy xxxxxxxx (xxxxxx, xxxxxxxx, xxxxxxxx). Xxx zvolit xxxxx xxxxx xxxxxxxxx. |
|
Xxxxxxx I.19. |
Číslo xxxxxxxxxx/xxxxxx: x&xxxx;xxxxxxxxxxx případech xxxxxxxxxxxx xxxxx. Xxxxx xxxxxxxxxx xxxx být xxxxxxx, xx-xx zboží xxxxxxxxxxxx x&xxxx;xxxxxxxxxx kontejnerech. Musí být xxxxxxx xxxxx číslo xxxxxx xxxxxx. Úřední xxxxxx xx použije x&xxxx;xxxxxxx, xx na xxxxxxxxx, nákladní xxxxxxx xxxx xxxxxxxxxx xxxxx xxxx připevněna plomba xxx xxxxxxxx příslušného xxxxxx, xxxxx xxxxxxxxx xxxxxx. |
|
Xxxxxxx I.20. |
Zboží xxxxxxxxx: xxxxxx zamýšlené xxxxxxx xxxxxxxx, xxx je xxxxxxxxxxxxx x&xxxx;xxxxxxxxxx xxxxxxx xxxxxxxxx Xxxxxxxx unie. K lidské xxxxxxxx: xxxx xx xxxxx xxxxxxxx určených x&xxxx;xxxxxx spotřebě. Krmivo: xxxx xx xxxxx xxxxxxxx xxxxxxxx xx xxxxxx xxxxxx. |
|
Xxxxxxx X.21. |
Xxxxxxxxx xx. |
|
Xxxxxxx X.22. |
Xxx xxxxxxx trh: xxx xxxxxxx zásilky xxxxxx x&xxxx;xxxxxxx xx xxx v Evropské xxxx. |
|
Xxxxxxx X.23. |
Xxxxxxx xxxxx balení: xxxxx xxxxxx. X&xxxx;xxxxxxx xxxxx ložených zásilek xx xxxx xxxxxxx xxxxxxxxx. |
|
Xxxxxxx X.24. |
Xxxxxxxx: Xxxxxxx xxxxx xxxxxxxx: tím xx xxxxxx hmotnost samotného xxxxx xxx xxxxxxxxxxxxxxx xxxxxx xxxx xxxxxx xxxxx. Xxxxxxx xxxxx hmotnost: xxxxxxx xxxxxxxx x&xxxx;xxxxxxxxxxx. Xxx se rozumí xxxxxxxx xxxxxxxx produktů x&xxxx;xxxxxxxxxxxxxx xxxxxx xx xxxxx obaly, xxxxx xxxxx xxxxxxxxxxx xxxxxxxxxx x&xxxx;xxxxxx přepravních xxxxxxxx. |
|
Xxxxxxx X.25. |
Xxxxx xxxxx: xxxxxx xxxxxxxxx xxx xxxxxxxxxxxxxxx xxxxxxx a název xxxxxx Xxxxxxxx xxxxx xxxxxxxxxx xxxxx xxxxxxxx Xxxx (XXX) č. 2658/87 (3). Tento xxxxx xxxxx xx x&xxxx;xxxxxxx xxxxxxx doplní x&xxxx;xxxxx xxxxxxxxx xxxxxxxx xx klasifikaci produktů. Uveďte xxxx, xxxx xxxxxxxx, xxxxx xxxxxx, xxxx xxxxx, číslo xxxxx, xxxxxx hmotnost a konečného xxxxxxxxxxxx, xxxxx xxxx xxxxxxxx xxxxxxxx pro xxxxxxxxx xxxxxxxxxxxx. Xxxx: vědecký xxxxx xxxx xxxxx xxxxxxxx v souladu x&xxxx;xxxxxxxx xxxxxxxx Xxxxxxxx xxxx. Xxxx xxxxx: xxxxxx xxxx xxxxx podle xxxxxxxx xxxxxxx x&xxxx;xxxxxxxxx X&xxxx;x&xxxx;XX xxxxxxxxxx x.&xxxx;21 XX/XXXXXX (Xxxxxxx XXX pro xxxxxxxxxxx obchodu x&xxxx;xxxxxxxxxxxx xxxxxxxxxxx). |
Xxxx XX: Xxxxxxxxx
Xxxx xxxx xxxx vyplnit xxxxxxxxxxx úředník xxxxxxxxx xxxxxxxxxx xxxxxxx třetí xxxx k podpisu xxxxxxxx xxxxxxxxx, jak je xxxxxxxxx x&xxxx;xx.&xxxx;88 xxxx.&xxxx;2 xxxxxxxx (EU) 2017/625.
|
Xxxxxxx XX. |
Xxxxx x&xxxx;xxxxxxxxx nezávadnosti: xxxx xxxx vyplňte x&xxxx;xxxxxxx se xxxxxxxxxx xxxxxxxxxxx xxxxxxxxx Xxxxxxxx xxxx týkajícími xx xxxxxx produktů, xxx xxxx xxxxxxxx v dohodách x&xxxx;xxxxxxxxxxxxx s určitými xxxxxxx xxxxxx nebo x&xxxx;xxxxxx xxxxxxxx xxxxxxxxxx Xxxxxxxx xxxx, xxxx xxxx xxxxxx xxxxxxxx x&xxxx;xxxxxxxxxxx. X&xxxx;xxxx XX.2.1, II.2.2, XX.2.3 x&xxxx;XX.2.4 xxxxxxx bod, xxxxx xxxxxxxx kategorii xxxxxxxx x&xxxx;xxxxxx (rizikům), xxx xxx se xxxxxxxxx vydává. Pokud xx xxxxxx xxxxxxxxx xxxxxxxxx xx XXXXX, osvědčující xxxxxxx prohlášení, xxxxx xxxxxx xxxxxxxxxx, xxxxxxxxx, xxxxxxxx x&xxxx;xxxxxxxxxx xxxx xx x&xxxx;xxxxxxxxx zcela xxxxxxxx. Xxxxx xx xxxxxxxxx xxxxxxx do IMSOC, xxxxxxxxxx, která nejsou xxxxxxxxxx, xx proškrtnou xxxx xx x&xxxx;xxxxxxxxx xxxxx xxxxxxxx. |
|
Xxxxxxx XX.x. |
Xxxxx xxxxxxx osvědčení: xxxxxx xxxxxxxxxx xxx xxxx x&xxxx;xxxxxxx X.2. |
|
Xxxxxxx II.b. |
Referenční xxxxx XXXXX: xxxxxx xxxxxxxxxx xxx xxxx x&xxxx;xxxxxxx X.2.x. Xxxxxxx xxxxx xxx úřední xxxxxxxxx vydaná x&xxxx;xxxxxxx XXXXX. |
|
Xxxxxxxxxxx xxxxxxx: |
Xxxxxxx příslušného xxxxxx xxxxx xxxx, xxxxx je xxxxx xxxxxxx xxxxxxxx xxxxxxxxxxx xxxxxx xxxxxxxxx: xxxxxx xxxxx (xxxxxxxx písmem), xxxxxxxxxxx a titul, xxxxxxxx xxxxxxxxxxxxx číslo x&xxxx;xxxxxxxxxx xxxxxxx xxxxxxxxxxx orgánu x&xxxx;xxxxx xxxxxxx. |
(1)&xxxx;&xxxx;Xxxxxx názvů xxxx a kódů: http://www.iso.org/iso/country_codes/iso-3166-1_decoding_table.htm.
(2) Nařízení Xxxx (XX) x.&xxxx;1/2005 xx xxx 22.&xxxx;xxxxxxxx&xxxx;2004 x&xxxx;xxxxxxx zvířat během xxxxxxxx x&xxxx;xxxxxxxxxxxxx činností x&xxxx;x&xxxx;xxxxx xxxxxxx 64/432/EHS x&xxxx;93/119/XX x&xxxx;xxxxxxxx (ES) x.&xxxx;1255/97 (Úř. xxxx. X&xxxx;3, 5.1.2005, s. 1).
(3) Nařízení Xxxx (XXX) x.&xxxx;2658/87 xx xxx 23. července 1987 x&xxxx;xxxxx x&xxxx;xxxxxxxxxxx xxxxxxxxxxxx x&xxxx;x&xxxx;xxxxxxxxx celním xxxxxxxxx (Xx. xxxx. X&xxxx;256, 7.9.1987, x.&xxxx;1).


